Advertisements
Advertisements
प्रश्न
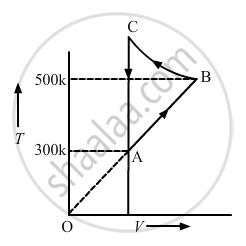
Consider the cyclic process ABCA, shown in figure, performed on a sample of 2.0 mol of an ideal gas. A total of 1200 J of heat is withdrawn from the sample in the process. Find the work done by the gas during the part BC.

योग
Advertisements
उत्तर
Given:-
Number of moles of the gas, n = 2
∆Q = − 1200 J (Negative sign shows that heat is extracted out from the system)
∆U = 0 ..............(During cyclic process)
Using the first law of thermodynamics, we get
∆Q = ∆U + ∆W
⇒ −1200 = 0 + (WAB + WBC + WCA)
Since the change in volume of the system applies on line CA, work done during CA will be zero.
From the ideal gas equation, we get
PV = nRT
P∆V = nR∆T
W = P∆V = nR∆T
⇒ ∆Q = ∆U + ∆W
⇒ −1200 = nR∆T + WBC + 0
⇒ −1200 = 2 × 8.3 × 200 + WBC
WBC = − 400 × 8.3 − 1200
= − 4520 J
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
