Advertisements
Advertisements
प्रश्न
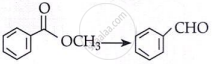
Complete the following reaction:
\[\ce{(C6H5CH2)2Cd + 2CH3COCI}\]
Advertisements
उत्तर
Treatment of acyl chlorides with dialkylcadmium gives a ketone.
\[\ce{2 CH3 - CO - CI + (C6H5- CH2)2 Cd -> \underset{\text{1-Phenylpropan-2-one}}{2 CH3-CO-CH2-C6H5} + CdCI2}\]
APPEARS IN
संबंधित प्रश्न
What is the action of Zn – Hg / conc. HCl on propanone?
How will you convert calcium acetate to acetaldehyde?
Answer the following in one sentence.
Predict the product of the following reaction:
\[\ce{CH3 - CH2 - COOCH3->[i) AIH(i-Bu)_2][ii) H3O+]}\]?
Which of the following alcohols needs acidic KMnO4 to convert it into aldehyde or ketone?
The reaction in which methyl group on benzene ring is converted to aldehydic group is called ______.
Identify 'A' and 'B' respectively in following reaction.
\[\ce{Toluene ->[Cl2][hv] A ->[H2O][\Delta] B}\]
The product of the reaction between dimethylcadmium and acetyl chloride is ____________.
Identify the compound A and Bin following reaction.
\[\ce{CH3Cl ->[KCN(alc)][\Delta] A ->[2H2O][HCl] B + NH4Cl}\]
Which of the following on heating with aqueous KOH produces acetaldehyde.
Identify the suitable reagent for the following conversion.