Advertisements
Advertisements
प्रश्न
A sample of air weighing 1.18 g occupies 1.0 × 103 cm3 when kept at 300 K and 1.0 × 105 Pa. When 2.0 cal of heat is added to it at constant volume, its temperature increases by 1°C. Calculate the amount of heat needed to increase the temperature of air by 1°C at constant pressure if the mechanical equivalent of heat is 4.2 × 107 erg cal−1. Assume that air behaves as an ideal gas.
Advertisements
उत्तर
Here,
m = 1.18 g = 1.18×10-3 kg
ΔQ = 2.0×4.2 J
P = 1.0×106 Pa
V = 1.0×103 cm3 = 1.0×10-3 m3
T = 300K
Applying eqn. of state
PV = nRT
=> n = PV/RT
=> n = 1.0×105×1.0×10-3/(8.314×300)
=> n = 0.04
ΔT = 10C
(ΔH)v = nCv ΔT
2.0×4.2 = nCv×1
=> Cv = 8.4/n= 8.4/0.04
=> Cv = 210
Again we know
Cp – Cv =R
=> Cp = R + Cv
=> Cp = 8.3 + 210
=> Cp = 218.3
Now at constant pressure
(ΔH)p = nCp ΔT
=> (ΔH)p = 218.3×0.04×1 = 8.732 J
In calories
=> (ΔH)p = 8.732/4.2 = 2.08 cal
APPEARS IN
संबंधित प्रश्न
A metre long narrow bore held horizontally (and closed at one end) contains a 76 cm long mercury thread, which traps a 15 cm column of air. What happens if the tube is held vertically with the open end at the bottom?
Can we define specific heat capacity for an adiabatic process?
Can a process on an ideal gas be both adiabatic and isothermal?
Show that the slope of the p−V diagram is greater for an adiabatic process compared to an isothermal process.
Can two states of an ideal gas be connected by an isothermal process as well as an adiabatic process?
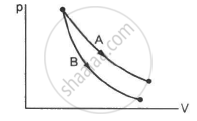
Two samples A and B are initially kept in the same state. Sample A is expanded through an adiabatic process and the sample B through an isothermal process. The final volumes of the samples are the same. The final pressures in A and B are pA and pBrespectively.
Let ∆Wa and ∆Wb be the work done by the systems A and B, respectively, in the previous question.
Consider the processes A and B shown in the figure. It is possible that
5 g of a gas is contained in a rigid container and is heated from 15°C to 25°C. Specific heat capacity of the gas at constant volume is 0.172 cal g−1 °C−1 and the mechanical equivalent of heat is 4.2 J cal−1. Calculate the change in the internal energy of the gas
A mixture contains 1 mole of helium (Cp = 2.5 R, Cv = 1.5 R) and 1 mole of hydrogen (Cp= 3.5 R, Cv = 2.5 R). Calculate the values of Cp, Cv and γ for the mixture.
In Joly's differential steam calorimeter, 3 g of an ideal gas is contained in a rigid closed sphere at 20°C. The sphere is heated by steam at 100°C and it is found that an extra 0.095 g of steam has condensed into water as the temperature of the gas becomes constant. Calculate the specific heat capacity of the gas in J g−1 K−1. The latent heat of vaporisation of water = 540 cal g−1
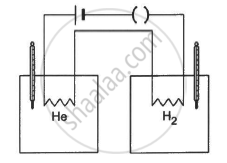
The figure shows two vessels with adiabatic walls, one containing 0.1 g of helium (γ = 1.67, M = 4 g mol−1) and the other containing some amount of hydrogen (γ = 1.4, M = 2 g mol−1). Initially, the temperatures of the two gases are equal. The gases are electrically heated for some time during which equal amounts of heat are given to the two gases. It is found that the temperatures rise through the same amount in the two vessels. Calculate the mass of hydrogen.
4.0 g of helium occupies 22400 cm3 at STP. The specific heat capacity of helium at constant pressure is 5.0 cal K−1 mol−1. Calculate the speed of sound in helium at STP.
Standing waves of frequency 5.0 kHz are produced in a tube filled with oxygen at 300 K. The separation between the consecutive nodes is 3.3 cm. Calculate the specific heat capacities Cp and Cv of the gas.
An engine takes in 5 moles of air at 20°C and 1 atm, and compresses it adiabatically to `1/10^"th"` of the original volume. Assuming air to be a diatomic ideal gas made up of rigid molecules, the change in its internal energy during this process comes out to be X kJ. The value of X to the nearest integer is ______.
If at same temperature and pressure, the densities for two diatomic gases are respectively d1 and d2 then the ratio of velocities of sound in these gases will be ______.
