Advertisements
Advertisements
प्रश्न
A capillary tube of radius 0.50 mm is dipped vertically in a pot of water. Find the difference between the pressure of the water in the tube 5.0 cm below the surface and the atmospheric pressure. Surface tension of water = 0.075 N m−1.
Advertisements
उत्तर
Given:
Radius of capillary tube r = 0.5 mm = 5 × 10−4 m
Depth (where pressure is to be found) h = 5.0 cm = 5 × 10−2 m
Surface tension of water T = 0.075 N/m
Excess pressure at 5 cm before the surface:
P = ρhg = 1000 × (5 × 10−2) × 9.8 = 490 N/m2
Excess pressure at the surface is given by:
\[P_0 = \frac{2T}{r} = \frac{2 \times \left( 0 . 75 \right)}{\left( 5 \times {10}^{- 4} \right)}\]
\[ = 300 \text{ N/ m}^2\]
Difference in pressure: P0 − P
\[= 490 - 300 = 190 \text{ N/ m}^2\]
Hence, the required difference in pressure is 190 N/m2.
APPEARS IN
संबंधित प्रश्न
A raindrop of diameter 4 mm is about to fall on the ground. Calculate the pressure inside the raindrop. [Surface tension of water T = 0.072 N/m, atmospheric pressure = 1.013 x 105 N/m2 ]
State any two characteristics of the angle of contact
Calculate the work done in increasing the radius of a soap bubble in air from 1 cm to 2 cm. The surface tension of soap solution is 30 dyne/cm. (Π = 3.142).
Water near the bed of a deep river is quiet while that near the surface flows. Give reasons.
The contact angle between a solid and a liquid is a property of
(a) the material of the solid
(b) the material of the liquid
(c) the shape of the solid
(d) the mass of the solid
A barometer is constructed with its tube having radius 1.0 mm. Assume that the surface of mercury in the tube is spherical in shape. If the atmospheric pressure is equal to 76 cm of mercury, what will be the height raised in the barometer tube? The contact angle of mercury with glass = 135° and surface tension of mercury = 0.465 N m−1. Density of mercury = 13600 kg m−3.
Solve the previous problem if the lead piece is fastened on the top surface of the block and the block is to float with its upper surface just dipping into water.
Define surface tension.
What will be the shape of the liquid meniscus for the obtuse angle of contact?
Numerical Problem.
A stone weighs 500 N. Calculate the pressure exerted by it if it makes contact with a surface of area 25 cm2.
How does surface tension help a plant?
What are the factors affecting the surface tension of a liquid?
A capillary of diameter d mm is dipped in water such that the water rises to a height of 30 mm. If the radius of the capillary is made `(2/3)` of its previous value, then compute the height up to which water will rise in the new capillary?
If the surface tension of a soap solution is 3 × 10-2 N/m then the work done in forming a soap film of 20 cm × 5 cm will be ______.
The excess of pressure, due to surface tension, on a spherical liquid drop of radius 'R' is proportional to ______.
Surface tension is exhibited by liquids due to force of attraction between molecules of the liquid. The surface tension decreases with increase in temperature and vanishes at boiling point. Given that the latent heat of vaporisation for water Lv = 540 k cal kg–1, the mechanical equivalent of heat J = 4.2 J cal–1, density of water ρw = 103 kg l–1, Avagadro’s No NA = 6.0 × 1026 k mole–1 and the molecular weight of water MA = 18 kg for 1 k mole.
- Estimate the energy required for one molecule of water to evaporate.
- Show that the inter–molecular distance for water is `d = [M_A/N_A xx 1/ρ_w]^(1/3)` and find its value.
- 1 g of water in the vapor state at 1 atm occupies 1601 cm3. Estimate the intermolecular distance at boiling point, in the vapour state.
- During vaporisation a molecule overcomes a force F, assumed constant, to go from an inter-molecular distance d to d ′. Estimate the value of F.
- Calculate F/d, which is a measure of the surface tension.
A soap film of surface tension 3 × 10-2 formed in a rectangular frame can support a straw as shown in Fig. If g = 10 ms-12, the mass of the straw is ______.
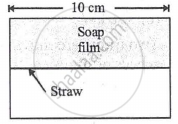
The surface tension of boiling water is ______.
A drop of water of radius 8 mm breaks into number of droplets each of radius 1 mm. How many droplets will be formed?
A light metal disc of radius ‘r’ floats on water surface and bends the surface downwards along the perimeter making an angle ‘θ’ with the vertical edge of the disc. If the weight of water displaced by the disc is ‘W’, the weight of the metal disc is ______.
[T = surface tension of water]
