Advertisements
Advertisements
Band theory of metals is based on ______
Concept: undefined >> undefined
Cyclopentadienyl anion is ______.
Concept: undefined >> undefined
Advertisements
Carboxylic acid reacts with diazomethane to yield:
Concept: undefined >> undefined
BF3 is a planar and electron-deficient compound. Hybridization and the number of electrons around the central atom, respectively are ______.
Concept: undefined >> undefined
The CORRECT statement amongst the following is:
Concept: undefined >> undefined
On heating the mixture of sodium benzoate and soda-lime, the product obtained is ______.
Concept: undefined >> undefined
Preparation of benzene from phenol is:
Concept: undefined >> undefined
Maximum decarboxylation occurs in:
Concept: undefined >> undefined
Electron occupies the available orbital singly before pairing in any one orbital occurs, it is ______.
Concept: undefined >> undefined
60 g of a compound on analysis gave 24 g C, 4 g H and 32 g O. The empirical formula of the compound is ______.
Concept: undefined >> undefined
Choose an INCORRECT statement about the electrophilic substitution reaction mechanism from the following:
Concept: undefined >> undefined
The number of electron pairs involved in the formation · of hydrogen cyanide molecule are
Concept: undefined >> undefined
The correct relationship between free energy change in a reaction and the corresponding equilibrium constant Kc is:
Concept: undefined >> undefined
Sodium salts of ______ acid are used as food preservative.
Concept: undefined >> undefined
At constant volume, 4 mol of an ideal gas when heated form 300 K to 500 K changes its internal energy by 5000 J. The molar heat capacity at constant volume is ______.
Concept: undefined >> undefined
What mass of CaCl2 in grams would be enough to produce 14.35 gm of AgCl?
Concept: undefined >> undefined
The IUPAC name of the following compound is ______.
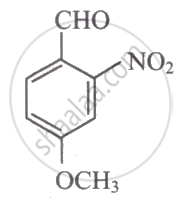
Concept: undefined >> undefined
The atomic number of elements is 17. The number of orbitals containing electron pairs in its valence shell is ______.
Concept: undefined >> undefined
For the given alkane,

The IUPAC name is:
Concept: undefined >> undefined
The dissociation constant value of four acids are as follows. Which of the values stand for strongest acid?
Concept: undefined >> undefined
Advertisements
Advertisements
| National Testing Agency NEET (UG) Medical Entrance Exam Question Bank Solutions |
|---|
| Question Bank Solutions for National Testing Agency NEET (UG) Medical Entrance Exam Biology |
| Question Bank Solutions for National Testing Agency NEET (UG) Medical Entrance Exam Chemistry |
| Question Bank Solutions for National Testing Agency NEET (UG) Medical Entrance Exam Physics |
