Advertisements
Advertisements
Why haloarenes are Less reactive than halo alkanes?
Concept: undefined >> undefined
Give the steps to understand the metal-ligand bonding.
Concept: undefined >> undefined
Advertisements
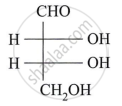
Assign D/L configuration to the following monosaccharides:
Concept: undefined >> undefined
Write the rate law for the following reaction:
A reaction that is zero order in A and second order in B.
Concept: undefined >> undefined
Write the rate law for the following reaction:
A reaction that is second order in NO and first order in Br2.
Concept: undefined >> undefined
For a chemical reaction A → 7 products, the rate of reaction doubles when the concentration of A is increased by a factor 4. The order of the reaction is ______.
Concept: undefined >> undefined
What are strong electrolytes? Give example.
Concept: undefined >> undefined
Predict the products (name and structure) in the following reaction:
\[\ce{CH3CH2CN ->[\Delta][dil. HCl]}\] ?
Concept: undefined >> undefined
Predict the products (name and structure) in the following reaction:
\[\ce{CH3 - CONH2 ->[\Delta][dil. HCl]}\] ?
Concept: undefined >> undefined
Predict the products (name and structure) in the following reaction:
\[\ce{C6H5 - CH2 - CH3 ->[alk. KMnO4][\Delta]}\] ?
Concept: undefined >> undefined
The rate constant of a reaction ______.
Concept: undefined >> undefined
What is thermodynamic equilibrium?
Concept: undefined >> undefined
Describe the action of the following reagent on glucose:
Bromine water
Concept: undefined >> undefined
Describe the action of the following reagent on glucose:
dil. Nitric acid.
Concept: undefined >> undefined
What is a state function?
Concept: undefined >> undefined
Which of the following is an extensive property?
Concept: undefined >> undefined
Wool is ______ fibre.
Concept: undefined >> undefined
Carbohydrates that do not undergo hydrolysis further are called ______.
Concept: undefined >> undefined
The pH of 0.0001 M of monobasic acid is ______.
Concept: undefined >> undefined
Diethyl ketone is an example of ______.
Concept: undefined >> undefined
