Advertisements
Advertisements
The rate constant for the first-order decomposition of H2O2 is given by the following equation:
`logk=14.2-(1.0xx10^4)/TK`
Calculate Ea for this reaction and rate constant k if its half-life period be 200 minutes.
(Given: R = 8.314 JK–1 mol–1)
Concept: undefined >> undefined
The rate constant of a first order reaction increases from 2 × 10−2 to 4 × 10−2 when the temperature changes from 300 K to 310 K. Calculate the energy of activation (Ea).
(log 2 = 0.301, log 3 = 0.4771, log 4 = 0.6021)
Concept: undefined >> undefined
Advertisements
What type of battery is mercury cell? Why is it more advantageous than dry cell?
Concept: undefined >> undefined
An element with density 2.8 g cm–3 forms a f.c.c. unit cell with edge length 4 x 10–8 cm. Calculate the molar mass of the element.
(Given : NA = 6.022 x 1023 mol –1)
Concept: undefined >> undefined
Name the type of cell which was used in Apollo space programme for providing electrical power.
Concept: undefined >> undefined
Define the following terms: Secondary batteries
Concept: undefined >> undefined
Which of the following pairs is an intensive property?
(A) Density, viscosity
(B) Surface tension, mass
(C) Viscosity, internal energy
(D) Heat capacity, volume
Concept: undefined >> undefined
In the Arrhenius equation for a first order reaction, the values of ‘A’ of ‘Ea’ are 4 × 1013 sec−1 and 98.6 kJ mol−1 respectively. At what temperature will its half life period be 10 minutes?
[R = 8.314 J K−1 mol−1]
Concept: undefined >> undefined
Which of the following is a basic oxide?
(A) SiO2
(B) P4O10
(C) MgO
(D) Al2O3
Concept: undefined >> undefined
55 L atm of work is obtained when 1.0 mole of an ideal gas is compressed isothermally from
a volume of 28.5 L to 18.5 L, the constant external pressure is
(A) 5.05 atm
(B) 5.5 atm
(C) 0.05 atm
(D) 0.55 atm
Concept: undefined >> undefined
A metal crystallises into two cubic faces namely face centered (FCC) and body centered (BCC), whose unit cell edge lengths are 3.5 Å and 3.0 Å respectively. Find the ratio of the densities of FCC and BCC.
Concept: undefined >> undefined
When phenol is heated with conc. HNO3 in presence of conc. H2SO4 it yields _______.
(A) o-nitrophenol
(B) p-nitrophenol
(C) 2,4,6-trinitrophenol
(D) m-nitrophenol
Concept: undefined >> undefined
Draw the structure of the chromate ion.
Concept: undefined >> undefined
Which of the following compounds is used as a semipermeable membrane?
(a) Potassium ferrocyanide
(b) Potassium argentocyanide
(c) Sodium meta aluminate
(d) Copper ferrocyanide
Concept: undefined >> undefined
The rate constant of a first order reaction are 0.58 S-1 at 313 K and 0.045 S-1 at 293 K. What is the energy of activation for the reaction?
Concept: undefined >> undefined
Predict the co-ordination No. of cs+ ion if `r_(Cs)^+` = 1.69Å and `r_(Cl)^-` = 1.81Å.
Concept: undefined >> undefined
IUPAC name of the following compound is
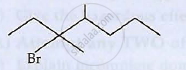
(a) 3 - Bromo- 3, 4- dimethylheptane
(b) 3, 4- dimethyl - 3- bromoheptane
(c) 5- Bromo- 4, 5- dimethylheptane
(d) 4, 5- dimethyl- 5- bromoheptane
Concept: undefined >> undefined
Write IUPAC names of the following compounds

Concept: undefined >> undefined
Write IUPAC names of the following compounds:

Concept: undefined >> undefined
