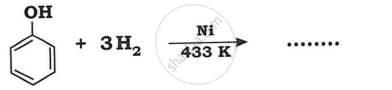
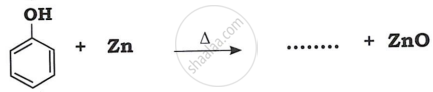
Advertisements
Advertisements
Write the name and formula of oxoacid of fluorine.
Concept: undefined >> undefined
Name two kinds of nucleic acids.
Concept: undefined >> undefined
Advertisements
Write the expression for work done in a chemical reaction.
Concept: undefined >> undefined
______ is a gaseous element of group 16.
Concept: undefined >> undefined
Inner transition elements are placed in ______ period.
Concept: undefined >> undefined
Write the ring structure of glucose.
Concept: undefined >> undefined
Identify A and B:
\[\ce{C6H5CH2Br ->[Alc.][KCN] A ->[Na/Ethanol] B}\]
Concept: undefined >> undefined
The transition elements are placed in period ______.
Concept: undefined >> undefined
What is the rate law for reaction
\[\ce{2NO_{2(g)}-> 2NO_{(g)} + O_{2(g)}}\]
Concept: undefined >> undefined
Write the correct molecular formula of gypsum.
Concept: undefined >> undefined
What are the nucleotides?
Concept: undefined >> undefined
Discuss the trends in electronegativity and atomic radii for elements of group 16 17, 18.
Concept: undefined >> undefined
Write two uses of coordination compounds.
Concept: undefined >> undefined
CH2 OH - CO - (CHOH)4 - CH2 OH is an example of ______.
Concept: undefined >> undefined
Identify the chiral molecule from the following.
Concept: undefined >> undefined
CH2OH-CO-(CHOH)4-CH2OH is an example of ______.
Concept: undefined >> undefined
The linkage present in Lactose is ______.
Concept: undefined >> undefined