Advertisements
Advertisements
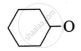
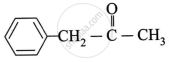
Write the structure of the products obtained from the following ketones by action of hydrazine in presence of strong base KOH.
Concept: undefined >> undefined
Arrange the following solutions in the order of increasing osmotic pressure (π) assuming complete ionization.
- 0.5M Li2 SO4
- 0.5M KCl
- 0.5M Al2 (SO4)3
- 0.1 M BaCl2
Concept: undefined >> undefined
Advertisements
In a particular reaction, 2 kJ of heat is released by the system and 8 kJ of work is done on the system. Determine ΔU.
Concept: undefined >> undefined
Identify B in the following reaction:
\[\begin{array}{cc}
\ce{O}\phantom{........}\\
||\phantom{........}\\
\ce{\underset{(Acetone)}{CH3 - C - CH3}->[CrO3] B}
\end{array}\]
Concept: undefined >> undefined
Calculate work done in oxidation of 4 moles of SO2 at 25°C. (Given: R = 8.314 JK−1 mol−1 ).
Concept: undefined >> undefined
E0cell (Standard Potential of cell) for the following cell reaction is ______.
\[\ce{Cd_{(s)} + Sn^{2+}_{(aq)}-> Cd^{2+}_{(aq)} + Sn_{(s)}}\]
(E0Cd = − 0.403 V and E0Sn = − 0.136 V)
Concept: undefined >> undefined
Which nanoparticles are used for purification of water?
Concept: undefined >> undefined
Write reactions when phenol reacts with Dilute HNO3
Concept: undefined >> undefined
Calculate pKa of HF if Ka= 7.2 x 10-4.
Concept: undefined >> undefined
Write names of two polyhalogen compounds.
Concept: undefined >> undefined
Write names of two polyhalogen compounds.
Concept: undefined >> undefined
Write two uses of polyhalogen compounds.
Concept: undefined >> undefined
Write two uses of polyhalogen compounds.
Concept: undefined >> undefined
Give the disadvantages of a vacancy defect.
Concept: undefined >> undefined
Mention the value of the bond angle in SO2.
Concept: undefined >> undefined
Convert the following Ethanal to ethanol.
Concept: undefined >> undefined
Convert the following iodomethane to methoxy methane.
Concept: undefined >> undefined
Convert the following iodomethane to methoxy methane.
Concept: undefined >> undefined
Define reverse osmosis.
Concept: undefined >> undefined