Advertisements
Advertisements
Aldehydes are readily oxidised to yield carboxylic acids but ketones are inert to oxidation. Which is the most likely explanation regarding this difference in reactivity?
Concept: undefined >> undefined
The following compound is a/an ____________.
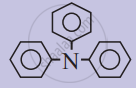
Concept: undefined >> undefined
Advertisements
The CORRECT relationship between molar solubility (S) and solubility product (Ksp) for salt, Cr(OH)3 is ____________.
Concept: undefined >> undefined
Which of the following is NOT an actinoid?
Concept: undefined >> undefined
In the reaction,
\[\ce{CH3COOH ->[SOCl2] X ->[Sodium salt of carboxylic acid] Y}\].
The compound Y was found to be a mixed acid anhydride. Thus, the sodium salt of carboxylic acid used CANNOT be ____________.
Concept: undefined >> undefined
Which is the CORRECT representation for the solubility product (Ksp) of CaF2?
Concept: undefined >> undefined
At constant temperature, a quantity of an ideal gas occupies 50 mL at 500 mmHg pressure. At what pressure, the volume will be 100 mL?
Concept: undefined >> undefined
Identify the element having highest enthalpy of atomisation from following.
Concept: undefined >> undefined
Which among the following lanthanoids, shows only +3 oxidation state?
Concept: undefined >> undefined
Which of the following compounds reacts with ammonia to form urotropine?
Concept: undefined >> undefined
Which of the following is a lanthanoid?
Concept: undefined >> undefined
Find INCORRECT statement with reference to NICAD cell.
Concept: undefined >> undefined
Concentration of the \[\ce{C2O^-_{4(aq)}}\] ions in a saturated solution of Ag2C2O4 is 1.1 × 10−4 mol L−1. Solubility product of Ag2C2O4 is __________.
Concept: undefined >> undefined
A cold drink bottle contains 200 mL liquid, in which CO2 is 0.1 molar. Considering CO2 as an ideal gas the volume of the dissolved CO2 at S.T.P is ______.
Concept: undefined >> undefined
In the reaction, \[\ce{C6H5COCH3 ->[{[H]}][Zn-Hg/conc. HCl] X}\], X is ______.
Concept: undefined >> undefined
The unit of ideal gas constant (R) is ____________.
Concept: undefined >> undefined
Identify the CORRECT statements from the following.
(I) Both lanthanoids and transition metals show magnetism.
(II) Both lanthanoids and actinoids form oxocations.
(III) Both lanthanoids and actinoids show decrease in ionic radius with an increase in atomic number.
(IV) Both lanthanoids and transition metals show fixed geometries.
Concept: undefined >> undefined
For an ideal gas, at constant temperature and pressure, the volume is ____________.
Concept: undefined >> undefined
Which is the gas evolved when carboxylic acids react with strongly electropositive metals (such as Na, K, Ca, Zn)?
Concept: undefined >> undefined
At a constant pressure, an ideal gas has a volume of 200 cm3 at 25°C. If the gas is cooled to −3°C, what will be the final volume of a gas?
Concept: undefined >> undefined
