Advertisements
Advertisements
The hardest substance among the following is:
Concept: undefined >> undefined
Molality of a solution relates the ____________.
Concept: undefined >> undefined
Advertisements
Molarity of the solution is ____________.
Concept: undefined >> undefined
The sum of all mole fraction for a mixture is always equal to ____________.
Concept: undefined >> undefined
Galvanic or a voltaic cell converts the chemical energy liberated during a redox reaction to ____________.
Concept: undefined >> undefined
Standard electrode potential is measured taking the concentrations of all the species involved in a half-cell is ____________.
Concept: undefined >> undefined
Standard hydrogen electrode operated under standard conditions of 1 atm H2 pressure, 298 K, and pH = 0 has a cell potential of ____________.
Concept: undefined >> undefined
For which of the following reaction the units of rate constant and rate of the reaction are same?
Concept: undefined >> undefined
The impurities associated with an ore is known as ____________.
Concept: undefined >> undefined
Which technique utilizes the density difference between the ore and the impurity to concentrate the ore is:
Concept: undefined >> undefined
The diagram shows a schematic representation of
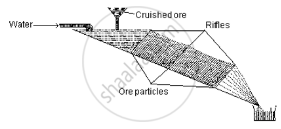
Concept: undefined >> undefined
In laboratory ammonia is prepared by heating:
Concept: undefined >> undefined
Ammonia has a higher boiling point and is less volatile because of ____________.
Concept: undefined >> undefined
Which compound is used in the preparation of caprolactam?
Concept: undefined >> undefined
Which compound is used as the cooling liquid in refrigerators?
Concept: undefined >> undefined
Ammonia act as a Lewis base because nitrogen has ____________.
Concept: undefined >> undefined
Which among the following elements does not belong to the first transition series?
Concept: undefined >> undefined
Which among the following is not considered as a part of transition elements?
Concept: undefined >> undefined
Electronic configuration of Cu3+ is ____________.
Concept: undefined >> undefined
Which of the following represents a chelate ligand?
Concept: undefined >> undefined
