Advertisements
Advertisements
Explain why does conductivity of germanium crystals increase on doping with galium.
Concept: undefined >> undefined
How does the doping increase the conductivity of semiconductors?
Concept: undefined >> undefined
Advertisements
Match the types of colloidal systems given in Column I with the name given in Column II.
| Column I | Column II |
| (i) Solid in liquid | (a) Foam |
| (ii) Liquid in solid | (b) Sol |
| (iii) Liquid in liquid | (c) Gel |
| (iv) Gas in liquid | (d) Emulsion |
Concept: undefined >> undefined
What is the role of adsorption in froth floatation process used especially for concentration of sulphide ores?
Concept: undefined >> undefined
Which of the following statements are correct?
(i) S – S bond is present in \[\ce{H2S2O6}\].
(ii) In peroxosulphuric acid \[\ce{(H2SO5)}\] sulphur is in +6 oxidation state.
(iii) Iron powder along with \[\ce{Al2O3}\] and \[\ce{K2O}\] is used as a catalyst in the preparation of \[\ce{NH3}\] by Haber’s process.
(iv) Change in enthalpy is positive for the preparation of \[\ce{SO3}\] by catalytic oxidation of \[\ce{SO2}\].
Concept: undefined >> undefined
Write the main product in the following reaction:
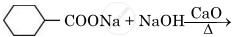
Concept: undefined >> undefined
Iron (z=26) is highly ferromagnetic. Explain.
Concept: undefined >> undefined
What type of magnetism is shown in the following alignment of magnetic moments?

Concept: undefined >> undefined
Account for the following: Fluorine does not exhibit positive oxidation state.
Concept: undefined >> undefined
Fluorine is a stronger oxidising agent than chlorine. Why?
Concept: undefined >> undefined
What type of magnetism is shown by a substance if magnetic moments of domains are arranged in same direction?
Concept: undefined >> undefined
What is the difference between a nucleoside and nucleotide?
Concept: undefined >> undefined
Compare the oxidizing action of F2 and Cl2 by considering parameters such as bond dissociation enthalpy, electron gain enthalpy and hydration enthalpy.
Concept: undefined >> undefined
Account for the following :
Acidic character increases from HF to HI.
Concept: undefined >> undefined
F2 has lower bond dissociation enthalpy than Cl2. Why?
Concept: undefined >> undefined
Define the following term:
Ferromagnetism
Concept: undefined >> undefined
Which halogen compound in the following pair will react faster in SN2 reaction CH3Br or CH3I.
Concept: undefined >> undefined
Which halogen compound in each of the following pairs will react faster in SN2 reaction
(CH3)3 C – Cl or CH3 – Cl
Concept: undefined >> undefined
Write the type of magnetism observed when the magnetic moments are oppositely aligned and cancel out each other.
Concept: undefined >> undefined
