Advertisements
Advertisements
When acidulated water (dil.H2SO4 solution) is electrolysed, will the pH of the solution be affected? Justify your answer.
Concept: undefined >> undefined
Write the cell reaction of a lead storage battery when it is discharged. How does the density of the electrolyte change when the battery is discharged?
Concept: undefined >> undefined
Advertisements
Why on dilution the m Λm of \[\ce{CH3COOH}\] increases very fast, while that of \[\ce{CH3COONa}\] increases gradually?
Concept: undefined >> undefined
Match the items of Column I and Column II on the basis of data given below:
`E_("F"_2//"F"^-)^Θ` = 2.87 V, `"E"_(("Li"^(+))//("Li"^-))^Θ` = − 3.5V, `"E"_(("Au"^(3+))//("Au"))^Θ` = 1.4 V, `"E"_(("Br"_(2))//("Br"^-))^Θ` = 1.09 V
| Column I | Column II |
| (i) F2 | (a) metal is the strongest reducing agent |
| (ii) Li | (b) metal ion which is the weakest oxidising agent |
| (iii) Au3+ | (c) non metal which is the best oxidising agent |
| (iv) Br– | (d) unreactive metal |
| (v) Au | (e) anion that can be oxidised by Au3+ |
| (vi) Li+ | (f) anion which is the weakest reducing agent |
| (vii) F– | (g) metal ion which is an oxidising agent |
Concept: undefined >> undefined
Assertion: Λm for weak electrolytes shows a sharp increase when the electrolytic solution is diluted.
Reason: For weak electrolytes degree of dissociation increases with dilution of solution.
Concept: undefined >> undefined
Assertion: `"E"_("Ag"^+ //"Ag")` increases with increase in concentration of Ag+ ions.
Reason: `"E"_("Ag"^+ //"Ag")` has a positive value.
Concept: undefined >> undefined
Assertion: Copper sulphate can be stored in zinc vessel.
Reason: Zinc is less reactive than copper.
Concept: undefined >> undefined
Consider figure and answer the question to given below.
How will the concentration of Zn2+ ions and Ag+ ions be affected after the cell becomes ‘dead’?
Concept: undefined >> undefined
Which of the following statements is incorrect about the collison theory of chemical reaction?
Concept: undefined >> undefined
Which of the following statements are applicable to a balanced chemical equation of an elementary reaction?
(i) Order is same as molecularity.
(ii) Order is less than the molecularity.
(iii) Order is greater than the molecularity.
(iv) Molecularity can never be zero.
Concept: undefined >> undefined
For a reaction A + B `->` Products, the rate law is — Rate = k [A][B]3/2 Can the reaction be an elementary reaction? Explain.
Concept: undefined >> undefined
Assertion: All collision of reactant molecules lead to product formation.
Reason: Only those collisions in which molecules have correct orientation and sufficient kinetic energy lead to compound formation.
Concept: undefined >> undefined
All energetically effective collisions do not result in a chemical change. Explain with the help of an example.
Concept: undefined >> undefined
What should be the considerations during the extraction of metals by electrochemical method?
Concept: undefined >> undefined
Which of the following acids forms three series of salts?
Concept: undefined >> undefined
Phosphorus forms a number of oxoacids. Out of these oxoacids phosphinic acid has strong reducing property. Write its structure and also write a reaction showing its reducing behaviour.
Concept: undefined >> undefined
Which of the following statements are correct about the reaction intermediate?

(i) Intermediate (c) is unstable because in this carbon is attached to 5 atoms.
(ii) Intermediate (c) is unstable because carbon atom is sp2 hybridised.
(iii) Intermediate (c) is stable because carbon atom is sp2 hybridised.
(iv) Intermediate (c) is less stable than the reactant (b).
Concept: undefined >> undefined
Draw other resonance structures related to the following structure and find out whether the functional group present in the molecule is ortho, para directing or meta directing.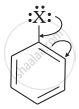
Concept: undefined >> undefined
Why is it necessary to avoid even traces of moisture during the use of a Grignard reagent?
Concept: undefined >> undefined
The process of converting alkyl halides into alcohols involves ______.
Concept: undefined >> undefined
