Advertisements
Advertisements
Dehydration of 2-butanol yields:
Concept: undefined >> undefined
Which of the following is not true in case of reaction with heated copper at 300°C?
Concept: undefined >> undefined
Advertisements
Lucas test is done to differentiate between ____________.
Concept: undefined >> undefined
Which one of the following on oxidation gives a ketone?
Concept: undefined >> undefined
Primary and secondary alcohols on the action of reduced copper give:
Concept: undefined >> undefined
The function of glucose is to ____________.
Concept: undefined >> undefined
The compound which gives the most stable carbonium ion on dehydration is:
Concept: undefined >> undefined
Cyclohexene is best prepared from cyclohexanol by which of the following:
Concept: undefined >> undefined
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
Concept: undefined >> undefined
\[\ce{CH3CH2OH}\] can be converted into \[\ce{CH3CHO}\] by ______.
Concept: undefined >> undefined
What would be the reactant and reagent used to obtain 2, 4-dimethyl pentan-3-ol?
Concept: undefined >> undefined
Identify the secondary alcohols from the following set:
- \[\ce{CH3CH2CH(OH)CH3}\]
- \[\ce{(C2H5)3COH}\]
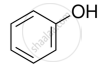
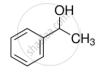
Concept: undefined >> undefined
Two beakers of capacity 500 mL were taken. One of these beakers, labelled as “A”, was filled with 400 mL water whereas the beaker labelled “B” was filled with 400 mL of 2 M solution of NaCl. At the same temperature both the beakers were placed in closed containers of same material and same capacity as shown in figure.
At a given temperature, which of the following statement is correct about the vapour pressure of pure water and that of NaCl solution.
Concept: undefined >> undefined
For a binary ideal liquid solution, the variation in total vapour pressure versus composition of solution is given by which of the curves?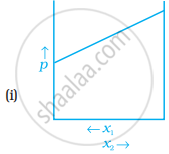

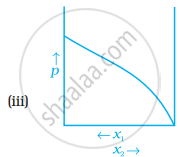
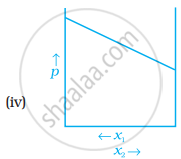
Concept: undefined >> undefined
Why is the vapour pressure of an aqueous solution of glucose lower than that of water?
Concept: undefined >> undefined
Which of the statements about solutions of electrolytes is not correct?
Concept: undefined >> undefined
\[\ce{\Lambda^0_m(NH4OH)}\] is equal to ______.
Concept: undefined >> undefined
\[\ce{Λ^0_m H2O}\] is equal to:
(i) \[\ce{Λ^0_m_{(HCl)} + \ce{Λ^0_m_{(NaOH)} - \ce{Λ^0_m_{(NaCl)}}}}\]
(ii) \[\ce{Λ^0_m_{(HNO_3)} + \ce{Λ^0_m_{(NaNO_3)} - \ce{Λ^0_m_{(NaOH)}}}}\]
(iii) \[\ce{Λ^0_{(HNO_3)} + \ce{Λ^0_m_{(NaOH)} - \ce{Λ^0_m_{(NaNO_3)}}}}\]
(iv) \[\ce{Λ^0_m_{(NH_4OH)} + \ce{Λ^0_m_{(HCl)} - \ce{Λ^0_m_{(NH_4Cl)}}}}\]
Concept: undefined >> undefined
Molar conductivity of ionic solution depends on:
(i) temperature.
(ii) distance between electrodes.
(iii) concentration of electrolytes in solution.
(iv) surface area of electrodes.
Concept: undefined >> undefined
Solutions of two electrolytes ‘A’ and ‘B’ are diluted. The Λm of ‘B’ increases 1.5 times while that of A increases 25 times. Which of the two is a strong electrolyte? Justify your answer.
Concept: undefined >> undefined
