Advertisements
Advertisements
Assertion (A): Sulphur present in an organic compound can be estimated quantitatively by Carius method.
Reason (R): Sulphur is separated easily from other atoms in the molecule and gets precipitated as light yellow solid.
Concept: undefined >> undefined
Assertion (A): Toluene on Friedal Crafts methylation gives o– and p–xylene.
Reason (R): CH3-group bonded to benzene ring increases electron density at o– and p– position.
Concept: undefined >> undefined
Advertisements
Define hydrogen bond. Is it weaker or stronger than the van der Waals forces?
Concept: undefined >> undefined
Discuss the principle of estimation of sulphur present in an organic compound.
Concept: undefined >> undefined
The ionisation constant of an acid, Ka, is the measure of strength of an acid. The Ka values of acetic acid, hypochlorous acid and formic acid are 1.74 × 10–5, 3.0 × 10–8 and 1.8 × 10–4 respectively. Which of the following orders of pH of 0.1 mol dm–3 solutions of these acids is correct?
Concept: undefined >> undefined
Discuss the principle of estimation of phosphorus present in an organic compound.
Concept: undefined >> undefined
Determine the solubilities of silver chromate, barium chromate, ferric hydroxide, lead chloride and mercurous iodide at 298K from their solubility product constants. Determine also the molarities of individual ions.
Concept: undefined >> undefined
What is the maximum concentration of equimolar solutions of ferrous sulphate and sodium sulphide so that when mixed in equal volumes, there is no precipitation of iron sulphide? (For iron sulphide, Ksp = 6.3 × 10–18).
Concept: undefined >> undefined
The ionization constant of nitrous acid is 4.5 × 10–4. Calculate the pH of 0.04 M sodium nitrite solution and also its degree of hydrolysis.
Concept: undefined >> undefined
Hydrogen bonds are formed in many compounds e.g., \[\ce{H2O, HF, NH3}\]. The boiling point of such compounds depends to a large extent on the strength of hydrogen bond and the number of hydrogen bonds. The correct decreasing order of the boiling points of above compounds is ______.
Concept: undefined >> undefined
In which of the following substances will hydrogen bond be strongest?
Concept: undefined >> undefined
Structures of molecules of two compounds are given below :
 |
 |
| (I) | (II) |
- Which of the two compounds will have intermolecular hydrogen bonding and which compound is expected to show intramolecular hydrogen bonding.
- The melting point of a compound depends on, among other things, the extent of hydrogen bonding. On this basis explain which of the above two compounds will show higher melting point.
- Solubility of compounds in water depends on power to form hydrogen bonds with water. Which of the above compounds will form hydrogen bond with water easily and be more soluble in it.
Concept: undefined >> undefined
Arrange the following in increasing order of \[\ce{pH}\].
\[\ce{KNO3 (aq), CH3COONa (aq), NH4Cl (aq), C6H5COONH4 (aq)}\]
Concept: undefined >> undefined
Consider structures I to VII and answer the question:
| I. | CH3 – CH2 – CH2 – CH2 – OH |
| II. | \[\begin{array}{cc} \ce{CH3 - CH2 - CH - CH3}\\ \phantom{.....}|\\ \phantom{.......}\ce{OH} \end{array}\] |
| III. | \[\begin{array}{cc} \phantom{...}\ce{CH3}\\ \phantom{}|\\ \ce{CH3 - C - CH3}\\ \phantom{}|\\ \phantom{..}\ce{OH} \end{array}\] |
| IV. | \[\begin{array}{cc} \ce{CH3 - CH - CH2 - OH}\\ |\phantom{........}\\ \ce{CH3}\phantom{......} \end{array}\] |
| V. | CH3 – CH2 – O – CH2 – CH3 |
| VI. | CH3 – O – CH2 – CH2 – CH3 |
| VII. | \[\begin{array}{cc} \ce{CH3 - O - CH - CH3}\\ \phantom{...}|\\ \phantom{......}\ce{CH3} \end{array}\] |
Which of the above compounds form pairs of metamers?
Concept: undefined >> undefined
The ionization constant of benzoic acid is 6.46 × 10–5 and Ksp for silver benzoate is 2.5 × 10–13. How many times is silver benzoate more soluble in a buffer of pH 3.19 compared to its solubility in pure water?
Concept: undefined >> undefined
Number of π bonds and σ bonds in the following structure is ______.
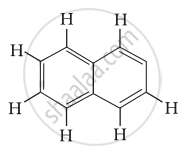
Concept: undefined >> undefined
Which molecule/ion out of the following does not contain unpaired electrons?
Concept: undefined >> undefined
Which of the following attain the linear structure?
(i) \[\ce{BeCl2}\]
(ii) \[\ce{NCO+}\]
(iii) \[\ce{NO2}\]
(iv) \[\ce{CS2}\]
Concept: undefined >> undefined
Discuss the significance/ applications of dipole moment.
Concept: undefined >> undefined
Which of the following will produce a buffer solution when mixed in equal volumes?
Concept: undefined >> undefined
