Advertisements
Advertisements
Atmospheric pressures recorded in different cities are as follows:
| Cities | Shimla | Bangalore | Delhi | Mumbai |
| p in N/m2 | 1.01 × 105 | 1.2 × 105 | 1.02 × 105 | 1.21 × 105 |
Consider the above data and mark the place at which liquid will boil first.
Concept: undefined >> undefined
Which of the following changes decrease the vapour pressure of water kept in a sealed vessel?
(i) Decreasing the quantity of water
(ii) Adding salt to water
(iii) Decreasing the volume of the vessel to one-half
(iv) Decreasing the temperature of water
Concept: undefined >> undefined
Advertisements
Pressure exerted by saturated water vapour is called aqueous tension. What correction term will you apply to the total pressure to obtain pressure of dry gas?
Concept: undefined >> undefined
The variation of vapour pressure of different liquids with temperature is shown in figure.
(i) Calculate graphically boiling points of liquids A and B.
(ii) If we take liquid C in a closed vessel and heat it continuously. At what temperature will it boil?
(iii) At high altitude, atmospheric pressure is low (say 60 mm Hg). At what temperature liquid D boils?
(iv) Pressure cooker is used for cooking food at hill station. Explain in terms of vapour pressure why is it so?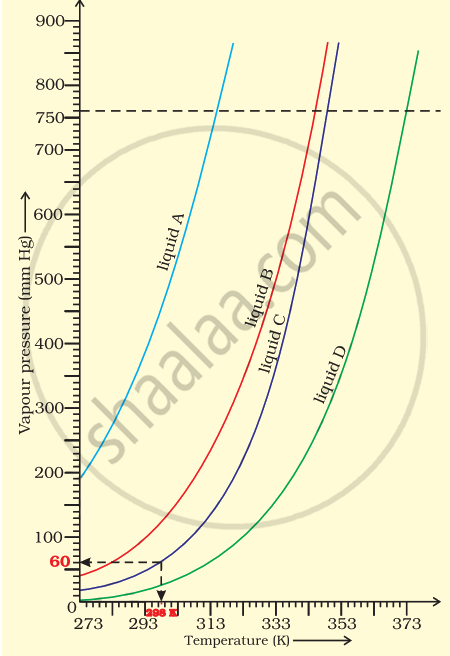
Concept: undefined >> undefined
Enthalpy of sublimation of a substance is equal to ______.
Concept: undefined >> undefined
Assertion (A): For any chemical reaction at a particular temperature, the equilibrium constant is fixed and is a characteristic property.
Reason (R): Equilibrium constant is independent of temperature.
Concept: undefined >> undefined
Electrophilic addition reactions proceed in two steps. The first step involves the addition of an electrophile. Name the type of intermediate formed in the first step of the following addition reaction.
\[\ce{H3C - HC = CH2 + H+ ->?}\]
Concept: undefined >> undefined
The addition of HCl to an alkene proceeds in two steps. The first step is the attack of H+ ion to  portion which can be shown as:
portion which can be shown as:
Concept: undefined >> undefined
Calculate the mass percent of different elements present in sodium sulphate (Na2SO4).
Concept: undefined >> undefined
Determine the empirical formula of an oxide of iron which has 69.9% iron and 30.1% dioxygen by mass.
Concept: undefined >> undefined
Determine the molecular formula of an oxide of iron in which the mass percent of iron and oxygen are 69.9 and 30.1, respectively.
Concept: undefined >> undefined
Using s, p, d notations, describe the orbital with the following quantum numbers n = 1, l = 0.
Concept: undefined >> undefined
Using s, p, d notations, describe the orbital with the following quantum numbers n = 3; l =1.
Concept: undefined >> undefined
Using s, p, d notations, describe the orbital with the following quantum numbers n = 4; l = 2.
Concept: undefined >> undefined
Using s, p, d notations, describe the orbital with the following quantum numbers n = 4; l =3.
Concept: undefined >> undefined
Calculate the enthalpy change for the process
\[\ce{CCl_4 (g) → C(g) + 4Cl(g)}\]
and calculate bond enthalpy of C–Cl in CCl4(g).
ΔvapHθ (CCl4) = 30.5 kJ mol–1.
ΔfHθ (CCl4) = –135.5 kJ mol–1.
ΔaHθ (C) = 715.0 kJ mol–1, where ΔaHθ is enthalpy of atomisation
ΔaHθ (Cl2) = 242 kJ mol–1
Concept: undefined >> undefined
The equilibrium constant expression for a gas reaction is,
`"K"_"c" = (["NH"_3]^4["O"_2]^5)/(["NO"]^4["H"_2"O"]^6)`
Write the balanced chemical equation corresponding to this expression.
Concept: undefined >> undefined
Arrange benzene, n-hexane and ethyne in decreasing order of acidic behaviour. Also give reason for this behaviour.
Concept: undefined >> undefined
Explain, why an organic liquid vaporises at a temperature below its boiling point in its steam distillation?
Concept: undefined >> undefined
Which of the following reactions will get affected by increasing the pressure? Also, mention whether change will cause the reaction to go into forward or backward direction.
- \[\ce{COCl2 (g) ⇌ CO (g) + Cl2 (g)}\]
- \[\ce{CH4 (g) + 2S2 (g) ⇌ CS2 (g) + 2H2S (g)}\]
- \[\ce{CO2 (g) + C (s) ⇌ 2CO (g)}\]
- \[\ce{2H2 (g) + CO (g) ⇌ CH3OH (g)}\]
- \[\ce{CaCO3 (s) ⇌ CaO (s) + CO2 (g)}\]
- \[\ce{4 NH3 (g) + 5O2 (g) ⇌ 4NO (g) + 6H2O(g)}\]
Concept: undefined >> undefined
