Advertisements
Advertisements
If travelling at same speeds, which of the following matter waves have the shortest wavelength?
Concept: undefined >> undefined
Which of the following will not show deflection from the path on passing through an electric field?
Concept: undefined >> undefined
Advertisements
A hypothetical electromagnetic wave is shown in Figure. Find out the wavelength of the radiation.
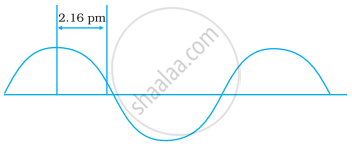
Concept: undefined >> undefined
What is photoelectric effect? State the result of photoelectric effect experiment that could not be explained on the basis of laws of classical physics. Explain this effect on the basis of quantum theory of electromagnetic radiations.
Concept: undefined >> undefined
Which of the following have no unit?
(i) Electronegativity
(ii) Electron gain enthalpy
(iii) Ionisation enthalpy
(iv) Metallic character
Concept: undefined >> undefined
Explain the following:
Electronegativity of elements increase on moving from left to right in the periodic table.
Concept: undefined >> undefined
If the electronic configuration of an element is 1s2 2s2 2p6 3s2 3p6 3d2 4s2, the four electrons involved in chemical bond formation will be ______.
Concept: undefined >> undefined
Why does type of overlap given in the following figure not result in bond formation?
 |
 |
Concept: undefined >> undefined
Briefly describe the valence bond theory of covalent bond formation by taking an example of hydrogen. How can you interpret energy changes taking place in the formation of dihydrogen?
Concept: undefined >> undefined
The pressure of a 1:4 mixture of dihydrogen and dioxygen enclosed in a vessel is one atmosphere. What would be the partial pressure of dioxygen?
Concept: undefined >> undefined
The enthalpies of elements in their standard states are taken as zero. The enthalpy of formation of a compound ______.
Concept: undefined >> undefined
Standard molar enthalpy of formation, ∆fHΘ is just a special case of enthalpy of reaction, ∆rHΘ. Is the ∆rHΘ for the following reaction same as ∆fHΘ? Give reason for your answer.
\[\ce{CaO(s) + CO2(g) -> CaCO3(s); ∆_fH^Θ = - 178.3 kJ mol^{-1}}\]
Concept: undefined >> undefined
The enthalpy of reaction for the reaction: \[\ce{2H2 (g) + O2(g) -> 2H2O (l)}\] is ∆rHΘ = – 572 kJ mol–1. What will be standard enthalpy of formation of \[\ce{H2O (l)}\]?
Concept: undefined >> undefined
Write a relation between ∆G and Q and define the meaning term and answer the following:
Why a reaction proceeds forward when Q < K and no net reaction occurs when Q = K.
Concept: undefined >> undefined
Write a relation between ∆G and Q and define the meaning term and answer the following:
Explain the effect of increase in pressure in terms of reaction quotient Q. for the reaction: \[\ce{CO (g) + 3H2 (g) ⇌ CH4 (g) + H2O (g)}\]
Concept: undefined >> undefined
Why does \[\ce{H+}\] ion always get associated with other atoms or molecules?
Concept: undefined >> undefined
Hydrogen peroxide is obtained by the electrolysis of ______.
Concept: undefined >> undefined
Which of the following statement(s) is/are correct in the case of heavy water?
(i) Heavy water is used as a moderator in nuclear reactor.
(ii) Heavy water is more effective as solvent than ordinary water.
(iii) Heavy water is more associated than ordinary water.
(iv) Heavy water has lower boiling point than ordinary water.
Concept: undefined >> undefined
How is heavy water prepared? Compare its physical properties with those of ordinary water.
Concept: undefined >> undefined
