Advertisements
Advertisements
Consider the following species:-
N3–, O2–, F–, Na+, Mg2+ and Al3+
Arrange them in the order of increasing ionic radii.
Concept: undefined >> undefined
Consider the following species:-
N3–, O2–, F–, Na+, Mg2+ and Al3+
What is common in them?
Concept: undefined >> undefined
Advertisements
Explain why cations are smaller and anions larger in radii than their parent atoms?
Concept: undefined >> undefined
The size of isoelectronic species — F-, Ne and Na+ is affected by ______.
Concept: undefined >> undefined
Explain the important aspects of resonance with reference to the `"CO"_3^(2-)` ion.
Concept: undefined >> undefined
H3PO3 can be represented by structures 1 and 2 shown below. Can these two structures be taken as the canonical forms of the resonance hybrid representing H3PO3? If not, give reasons for the same.
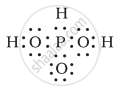 |
 |
| (1) | (2) |
Concept: undefined >> undefined
Write the resonance structure for SO3.
Concept: undefined >> undefined
Write the resonance structures for NO2.
Concept: undefined >> undefined
Write the resonance structures for `"NO"_3^(-)`.
Concept: undefined >> undefined
At 0°C, the density of a certain oxide of a gas at 2 bar is same as that of dinitrogen at 5 bar. What is the molecular mass of the oxide?
Concept: undefined >> undefined
The pressure of 1 g of an ideal gas A at 27 °C is found to be 2 bar. When 2 g of another ideal gas B is introduced in the same flask at the same temperature the pressure becomes 3 bar. Find a relationship between their molecular masses.
Concept: undefined >> undefined
The drain cleaner, Drainex contains small bits of aluminum which react with caustic soda to produce dihydrogen. What volume of dihydrogen at 20 °C and one bar will be released when 0.15g of aluminum reacts?
Concept: undefined >> undefined
The density of a gas is found to be 5.46 g/dm3 at 27 °C at 2 bar pressure. What will be its density at STP?
Concept: undefined >> undefined
How much time would it take to distribute one Avogadro number of wheat grains, if 1010 grains are distributed each second?
Concept: undefined >> undefined
Comment on the thermodynamic stability of NO(g), given
\[\ce{1/2 N_2 (g) + 1/2 O2(g) → NO(g)}\]; ΔrHθ = 90 kJ mol–1
\[\ce{NO(g) +1/2 O2(g) → NO2(g)}\] : ΔrHθ= –74 kJ mol–1
Concept: undefined >> undefined
Why is Wurtz reaction not preferred for the preparation of alkanes containing odd number of carbon atoms? Illustrate your answer by taking one example
Concept: undefined >> undefined
Ethyl acetate is formed by the reaction between ethanol and acetic acid and the equilibrium is represented as:
\[\ce{CH3COOH (l) + C2H5OH (l) ⇌ CH3COOC2H5 (l) + H2O (l)}\]
Write the concentration ratio (reaction quotient), Qc, for this reaction (note: water is not in excess and is not a solvent in this reaction).
Concept: undefined >> undefined
Ethyl acetate is formed by the reaction between ethanol and acetic acid and the equilibrium is represented as:
\[\ce{CH3COOH (l) + C2H5OH (l) ⇌ CH3COOC2H5 (l) + H2O (l)}\]
At 293 K, if one starts with 1.00 mol of acetic acid and 0.18 mol of ethanol, there is 0.171 mol of ethyl acetate in the final equilibrium mixture. Calculate the equilibrium constant.
Concept: undefined >> undefined
Ethyl acetate is formed by the reaction between ethanol and acetic acid and the equilibrium is represented as:
\[\ce{CH3COOH (l) + C2H5OH (l) ⇌ CH3COOC2H5 (l) + H2O (l)}\]
Starting with 0.5 mol of ethanol and 1.0 mol of acetic acid and maintaining it at 293 K, 0.214 mol of ethyl acetate is found after sometime. Has equilibrium been reached?
Concept: undefined >> undefined
Suggest a reason as to why CO is poisonous.
Concept: undefined >> undefined
