Advertisements
Advertisements
The transfer of energy in a food chain is always ______.
Concept: undefined >> undefined
If the real image of a candle flame formed by a lens is three times the size of the flame and the distance between lens and image is 80 cm, at what distance should the candle be placed from the lens?
Concept: undefined >> undefined
Advertisements
Observe the food web and answer the questions given below:
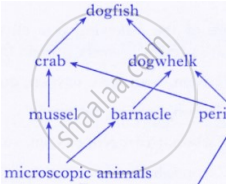
The mussel can be described as:
Concept: undefined >> undefined
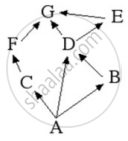
Observe the food web and answer the questions given below:

In the food web, what two organisms are competing for food?
Concept: undefined >> undefined
Name the type of mirror used in the following situation:
Side/rear-view mirror of a vehicle
Support your answer with reason.
Concept: undefined >> undefined
Name the type of mirror used in the following situation:
Solar furnace
Support your answer with reason.
Concept: undefined >> undefined
Solid calcium oxide reacts vigorously with water to form calcium hydroxide accompanied by liberation of heat. This process is called slaking of lime. Calcium hydroxide dissolves in water to form a solution called lime water. Which among the following is (are) true about slaking of lime and the solution formed?
- It is an endothermic reaction
- It is an exothermic reaction
- The pH of the resulting solution will be more than seven
- The pH of the resulting solution will be less than seven
Concept: undefined >> undefined
Barium chloride on reacting with ammonium sulphate forms barium sulphate and ammonium chloride. Which of the following correctly represents the type of the reaction involved?
- Displacement reaction
- Precipitation reaction
- Combination reaction
- Double displacement reaction
Concept: undefined >> undefined
Electrolysis of water is a decomposition reaction. The mole ratio of hydrogen and oxygen gases liberated during electrolysis of water is
Concept: undefined >> undefined
Which of the following is(are) an endothermic process(es)?
- Dilution of sulphuric acid
- Sublimation of dry ice
- Condensation of water vapours
- Evaporation of water
Concept: undefined >> undefined
In the double displacement reaction between aqueous potassium iodide .and aqueous lead nitrate, a yellow precipitate of lead iodide is formed. While performing the activity if lead nitrate is not available, which of the following can be used in place of lead nitrate?
Concept: undefined >> undefined
Which of the following gases can be used for storage of fresh sample of an oil for a long time?
Concept: undefined >> undefined
The following reaction is used for the preparation of oxygen gas in the laboratory
`2"KClO"_3("s") overset("Heat")underset("Catalyst")(->) 2"KCl"("s") + 3"O"_2 ("g")`
Which of the following statement(s) is(are) correct about the reaction ?
Concept: undefined >> undefined
Which one of the following processes involve chemical reactions?
Concept: undefined >> undefined
In which of the following chemical equations, the abbreviations represent the correct states of the reactants and products involved at reaction temperature?
Concept: undefined >> undefined
Which of the following are combination reactions?
- `2"KClO"_3 overset("Heat")(->) 2"KCl" + 3"O"_2`
- `"MgO" + "H"_2"O" -> "Mg"("OH")_2`
- `4"Al" + 3"O"_2 -> 2"Al"_2"O"_3`
- `"Zn" + "FeSO"_4 -> "ZnSO"_4 + "Fe"`
Concept: undefined >> undefined
Write the balanced chemical equations for the following reactions and identify the type of reaction in each case.
- Nitrogen gas is treated with hydrogen gas in the presence of a catalyst at 773 K to form ammonia gas.
- Potassium hydroxide solution is treated with acetic acid to form potassium acetate and water.
- Ethanol is warmed with ethanoic acid to form ethyl acetate in the presence of concentrated H2SO4.
- Ethene is burnt in the presence of oxygen to form carbon dioxide, water and release heat and light.
Concept: undefined >> undefined
Write the balanced chemical equations for the following reactions and identify the type of reaction in each case.
- Iron (III) oxide reacts with aluminum and gives molten iron and aluminum oxide.
- Magnesium ribbon is burnt in an atmosphere of nitrogen gas to form solid magnesium nitride.
- Chlorine gas is passed in an aqueous potassium iodide solution to form potassium chloride solution and solid iodine.
- Ethanol is burnt in air to form carbon dioxide, water and releases heat.
Concept: undefined >> undefined
Complete the missing component /variable given as x and y in the following reactions :
`"Pb"("NO"_3)_2("aq") + 2"Kl"("aq") -> "Pbl"_2(x) + 2"KNO"_3("y")`
Concept: undefined >> undefined
Complete the missing component /variable given as x and y in the following reactions
`"Cu"("s") + 2"AgNO"_3("aq") -> "Cu"("NO"_3)_2("aq") + x("s")`
Concept: undefined >> undefined
