Advertisements
Advertisements
Which of the following statements are usually correct for carbon compounds? These
- are good conductors of electricity
- are poor conductors of electricity
- have strong forces of attraction between their molecules
- do not have strong forces of attraction between their molecules.
Concept: undefined >> undefined
Cation is formed when ______.
Concept: undefined >> undefined
Advertisements
The I.U.P.A.C name of CH3CH2CH = CH2 is?
Concept: undefined >> undefined
Which of the following compounds of carbon does not consist of ions?
Concept: undefined >> undefined
Which of the following statements are correct for carbon compounds?
- Most carbon compounds are good conductors of electricity.
- Most carbon compounds are poor conductors of electricity.
- Force of attraction between molecules of carbon compounds is not very strong.
- Force of attraction between molecules of carbon compounds is very strong.
Concept: undefined >> undefined
Which of the following is the formula of Butanoic acid?
Concept: undefined >> undefined
Which of the following statement is not correct about the magnetic field?
Concept: undefined >> undefined
In a given food chain if the amount of energy at the fourth trophic level is 6 kJ, what will be the energy available at the producer level?
Concept: undefined >> undefined
We should reduce the use of the plastic bags, bottles etc. because ______.
Concept: undefined >> undefined
Green plants utilize ______ percent of sun’s energy to prepare their food by the process of photosynthesis.
Concept: undefined >> undefined
The process of accumulation of harmful chemical substances like pesticides, in the body of living organisms at each trophic level of a food chain is known as ______.
Concept: undefined >> undefined
If 100 J energy is available at the producer level in a food chain then the energy available to the secondary consumer will be ______.
Concept: undefined >> undefined
In an ecosystem, herbivores represent ______.
Concept: undefined >> undefined
A food chain comprising birds, green plants, fish and man. The concentration of harmful chemical entering the food chain will be maximum in ______.
Concept: undefined >> undefined
Which of the following is a logical sequence of food chain?
Concept: undefined >> undefined
Which of the following is an autotroph?
Concept: undefined >> undefined
Magnetic lines of force inside current carrying solenoid are ____________.
Concept: undefined >> undefined
Food chains are very important for the survival of most species.

If 10,000 J solar energy falls on green plants in a terrestrial ecosystem, what percentage of solar energy will be converted into food energy?
Concept: undefined >> undefined
Food chains are very important for the survival of most species.

Matter and energy are two fundamental inputs of an ecosystem. Movement of:
Concept: undefined >> undefined
Observe the food web and answer the questions given below:
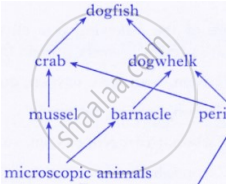
Why do all food chains start with plants?
Concept: undefined >> undefined
