Advertisements
Advertisements
Question
Write the significance and limitations of octet rule.
Long Answer
Advertisements
Solution 1
Limitations of the octet theory:
The following are the limitations of the octet rule:
- The rule failed to predict the shape and relative stability of molecules.
- It is based upon the inert nature of noble gases. However, some noble gases like xenon and krypton form compounds such as XeF2, KrF2 etc.
- The octet rule cannot be applied to the elements in and beyond the third period of the periodic table. The elements present in these periods have more than eight valence electrons around the central atom. For example: PF5, SF6, etc.

- The octet rule is not satisfied for all atoms in a molecule having an odd number of electrons. For example, NO and NO2 do not satisfy the octet rule.

- This rule cannot be applied to those compounds in which the number of electrons surrounding the central atom is less than eight. For example, LiCl, BeH2, AlCl3 etc. do not obey the octet rule.
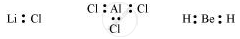
shaalaa.com
Solution 2
Significance: It helps to explain why different atoms combine with each other to form ionic compounds or covalent compounds.
Limitations of Octet rule:
- According to Octet rule, atoms take part in chemical combination to achieve the configuration of nearest noble gas elements. However, some of noble gas elements like Xenon have formed compounds with fluorine and oxygen. For example: XeF2, XeF4 etc.
Therefore, validity of the octet rule has been challenged. - This theory does not account for shape of molecules.
shaalaa.com
Is there an error in this question or solution?
