Advertisements
Advertisements
Question
Write a short note on Sandmeyer’s reaction.
Advertisements
Solution
Sandmeyer’s reaction:
When a primary aromatic amine, dissolved or suspended in aqueous mineral acid, is treated with sodium nitrite, a diazonium salt is formed. Mixing solution of freshly prepared diazonium salt with cuprous chloride or cuprous bromide, results in the replacement of the diazonium group by - Cl or - Br. This reaction is known as Sandmeyer' s reaction
eg.

APPEARS IN
RELATED QUESTIONS
How is propene converted into 1- bromopropane and 2 - bromopropane?
How do you convert the following: Chlorobenzene to 2-chlorotoluene
Write the structure of the major product in each of the following reaction :

\[\ce{CH3 = CH2CH3 + H - I -> CH3CH2CH2I + CH3CHICH3}\] (major). This reaction is:
The synthesis of alkyl fluorides is best accomplished by ____________.
The best method for the conversion of an alcohol into an alkyl chloride is by treating the alcohol with ____________.
The catalyst used in the preparation of an alkyl chloride by the action of dry HCl on alcohol is ____________.
Among the following, the most reactive towards alcoholic KOH is:
\[\ce{X ->[AgNO3][HNO3] Yellow or While ppt}\]
Which of the following cannot be X?
The order of reactivity of alcohols with halogen acids is ______.
(A) \[\ce{CH3CH2 - CH2 - OH}\]
(B) \[\begin{array}{cc}
\phantom{}\ce{CH3CH2 - CH - OH}\\
\phantom{...}\phantom{}|\\
\phantom{......}\ce{CH3}
\end{array}\]
(C) \[\begin{array}{cc}
\phantom{........}\ce{CH3}\\
\phantom{.....}\phantom{}|\\
\phantom{}\ce{CH3CH2 - C - OH}\\
\phantom{.....}\phantom{}|\\
\phantom{........}\ce{CH3}
\end{array}\]
Alkyl fluorides are synthesised by heating an alkyl chloride/bromide in presence of ______ or ______.
(i) CaF2
(ii) CoF2
(ii) Hg2F2
(iv) NaF
Identify the products A and B formed in the following reaction:
\[\ce{CH3 - CH2 - CH = CH - CH3 + HCl -> A + B}\]
A hydrocarbon of molecular mass 72 g mol–1 gives a single monochloro derivative and two dichloro derivatives on photo chlorination. Give the structure of the hydrocarbon.
Which of the following compounds would undergo SN1 reaction faster and why?
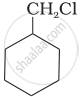 |
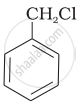 |
| (A) | (B) |
Match the structures given in Column I with the names in Column II.
| Column I | Column II | |
| (i) |  |
(a) 4-Bromopent-2-ene |
| (ii) | 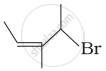 |
(b) 4-Bromo-3-methylpent-2-ene |
| (iii) | 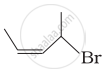 |
(c) 1-Bromo-2-methylbut-2-ene |
| (iv) | 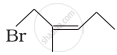 |
(d) 1-Bromo-2-methylpent-2-ene |
Some alkylhalides undergo substitution whereas some undergo elimination reaction on treatment with bases. Discuss the structural features of alkyl halides with the help of examples which are responsible for this difference.
Which of the following is the most stable free radical?
Which compound would undergo dehydrohalogenation with strong base to give the alkene shown below as the only alkene product?
CH3 – CH2CH = CH – CH3
The alkyl halide which does not give white precipitate with alcoholic AgNO3 solution is :-
\[\begin{array}{cc}
\ce{Ph - CH - CH2 - CH2 ->[Zn - Cu][Δ] Product}\\
\phantom{..}|\phantom{......}|\phantom{....................}\\
\phantom{}\ce{Br}\phantom{....}\ce{Br}\phantom{..................}
\end{array}\]
Product of the above reaction is
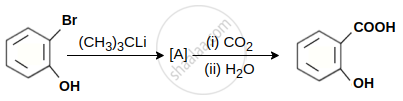
In the given conversion the compound A is:
In Finkelstein Reaction, which reactants are used?
The number of stereoisomers are possible for a compound of the molecular formula \[\ce{CH3 - CH = CH - CH(OH) - Me}\] is ______.
