Advertisements
Advertisements
Choose the most correct option.
The best method for the preparation of alkyl fluorides is _______.
Concept: undefined >> undefined
Butanenitrile may be prepared by heating ______.
Concept: undefined >> undefined
Advertisements
Choose the most correct option.
Choose the compound from the following that will react fastest by SN1 mechanism
Concept: undefined >> undefined
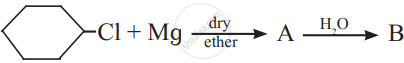
The product ‘B’ in the above reaction sequence is,
Concept: undefined >> undefined
Choose the most correct option.
Which of the following is likely to undergo racemization during alkaline hydrolysis?
(I)
\[\begin{array}{cc}
\ce{CH3-CH-C2H5}\\
|\\
\ce{Cl}\end{array}\]
(II)
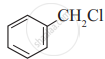
(III)
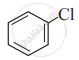
(IV)
\[\begin{array}{cc}
\ce{\phantom{.......}CH3}\\
\phantom{.....}|\\
\ce{CH3-CH}\\
\phantom{.....}|\\
\ce{\phantom{..........}CH2Cl}
\end{array}\]
Concept: undefined >> undefined
Match the pairs.
| Column I | Column II |
| \[\begin{array}{cc} \ce{CH3CH - CH3}\\ |\phantom{.....}\\ \ce{X\phantom{.....}} \end{array}\] |
vinyl halide |
| \[\ce{CH2 = CH - CH2X}\] | alkyl halide |
| \[\ce{CH2 = CH - X}\] | allyl halide |
| benzyl halide | |
| aryl halide |
Concept: undefined >> undefined
Choose the most correct option.
The hybridisation of nitrogen in primary amine is ____________.
Concept: undefined >> undefined
Isobutylamine is an example of ______.
Concept: undefined >> undefined
Choose the most correct option.
Which type of amine does produce N2 when treated with HNO2?
Concept: undefined >> undefined
Choose the most correct option.
Carbylamine test is given by ____________.
Concept: undefined >> undefined
Choose the most correct option.
Identify ‘B’ in the following reactions
\[\ce{CH3 - C ≡ N ->[Na/C2H5OH] A ->[NaNO2/dilHCl]B}\]
Concept: undefined >> undefined
\[\ce{CH2OH-CO-(CHOH)4-CH2OH}\] is an example of ______.
Concept: undefined >> undefined
Select the most correct choice.
Open chain formula of glucose does not contain ______.
Concept: undefined >> undefined
Which of the following does not apply to \[\ce{CH2NH2-COOH}\]?
Concept: undefined >> undefined
Select the most correct choice.
Tryptophan is called essential amino acid because
Concept: undefined >> undefined
Select the most correct choice.
A disulphide link gives rise to the following structure of a protein.
Concept: undefined >> undefined
Give scientific reasons:
The disaccharide sucrose gives negative Tollens test while the disaccharide maltose gives a positive Tollens test.
Concept: undefined >> undefined
Give scientific reasons:
On complete hydrolysis DNA gives equimolar quantities of adenine and thymine.
Concept: undefined >> undefined
Give scientific reasons:
α-Amino acids have high melting points compared to the corresponding amines or carboxylic acids of comparable molecular mass.
Concept: undefined >> undefined
