Advertisements
Advertisements
प्रश्न
Choose the most correct option.
Which of the following is likely to undergo racemization during alkaline hydrolysis?
(I)
\[\begin{array}{cc}
\ce{CH3-CH-C2H5}\\
|\\
\ce{Cl}\end{array}\]
(II)
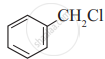
(III)
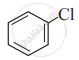
(IV)
\[\begin{array}{cc}
\ce{\phantom{.......}CH3}\\
\phantom{.....}|\\
\ce{CH3-CH}\\
\phantom{.....}|\\
\ce{\phantom{..........}CH2Cl}
\end{array}\]
पर्याय
Only I
Only II
II and IV
Only IV
Advertisements
उत्तर
Only I
APPEARS IN
संबंधित प्रश्न
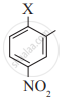
The correct order of increasing reactivity of C-X bond towards nucleophile in the following compounds is:
(I)
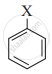
(II)
(III) (CH3)3 C-X
(IV) (CH3)2 CH-X
Choose the most correct option.
The best method for the preparation of alkyl fluorides is _______.
Butanenitrile may be prepared by heating ______.
Choose the most correct option.
Choose the compound from the following that will react fastest by SN1 mechanism
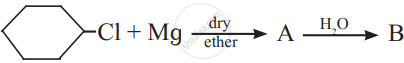
The product ‘B’ in the above reaction sequence is,
Match the pairs.
| Column I | Column II |
| \[\begin{array}{cc} \ce{CH3CH - CH3}\\ |\phantom{.....}\\ \ce{X\phantom{.....}} \end{array}\] |
vinyl halide |
| \[\ce{CH2 = CH - CH2X}\] | alkyl halide |
| \[\ce{CH2 = CH - X}\] | allyl halide |
| benzyl halide | |
| aryl halide |
The type of mono halogen derivative in which a halogen atom is bonded to sp3 hybridized carbon atom next to carbon-carbon double bond is ______________
The common name of 1-chloro-2, 2-dimethyl propane is ______.
When propene reacts with HCl in presence of peroxide, the product is ________.
The conversion of 2-methylpropan-1-ol to 2-methylpropan-2-ol is ______.
IUPAC name of isobutyl chloride is ______.
Alkyl chloride containing 4° carbon atom(s) would be ____________.
Which of the following compounds is obtained when t-butyl bromide is treated with alcoholic ammonia?
The number of possible monohalogen derivatives for the alkyl halide having molecular formula C4H9X is ____________.
What is the IUPAC name of neopentyl chloride?
Identify A and B respectively in the following conversion.
\[\ce{Ethene ->[A] Bromoethane ->[B][\Delta] Ethyl propionate}\]
The IUPAC name of isobutyl bromide is ____________.
The decreasing order of the rate of nitration of benzene (I), C6D6 (II), nitrobenzene (III), chlorobenzene (IV) is ______.
The allylic halide, among the following, is ______.
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry either] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Identify the chiral molecule from the following.
Identify the chiral molecule from the following.
Among the following vinylic halide is ______.
