Advertisements
Advertisements
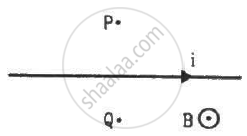
A long, straight wire carrying a current of 1.0 A is placed horizontally in a uniform magnetic field B = 1.0 × 10−5 T pointing vertically upward figure. Find the magnitude of the resultant magnetic field at the points P and Q, both situated at a distance of 2.0 cm from the wire in the same horizontal plane.
Concept: undefined >> undefined
A long, straight wire of radius r carries a current i and is placed horizontally in a uniform magnetic field B pointing vertically upward. The current is uniformly distributed over its cross section. (a) At what points will the resultant magnetic field have maximum magnitude? What will be the maximum magnitude? (b) What will be the minimum magnitude of the resultant magnetic field?
Concept: undefined >> undefined
Advertisements
A hypothetical magnetic field existing in a region is given by `vecB = B_0 vece` where `vece`_r denotes the unit vector along the radial direction. A circular loop of radius a, carrying a current i, is placed with its plane parallel to the x−y plane and the centre at (0, 0, d). Find the magnitude of the magnetic force acting on the loop.
Concept: undefined >> undefined
A straight wire of length l can slide on two parallel plastic rails kept in a horizontal plane with a separation d. The coefficient of friction between the wire and the rails is µ. If the wire carries a current i, what minimum magnetic field should exist in the space in order to slide the wire on the rails?
Concept: undefined >> undefined
The magnetic field existing in a region is given by `vecB = B_0(1 + x/1)veck` . A square loop of edge l and carrying a current i, is placed with its edges parallel to the x−y axes. Find the magnitude of the net magnetic force experienced by the loop.
Concept: undefined >> undefined
An electric kettle used to prepare tea, takes 2 minutes to boil 4 cups of water (1 cup contains 200 cc of water) if the room temperature is 25°C. (a) If the cost of power consumption is Re 1.00 per unit (1 unit = 1000 watt-hour), calculate the cost of boiling 4 cups of water. (b) What will be the corresponding cost if the room temperature drops to 5°C?
Concept: undefined >> undefined
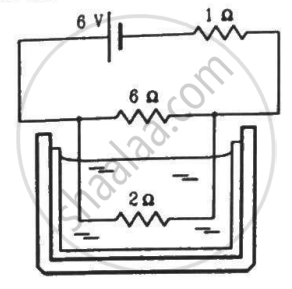
The 2.0 Ω resistor shown in the figure is dipped into a calorimeter containing water. The heat capacity of the calorimeter together with water is 2000 J K−1. (a) If the circuit is active for 15 minutes, what would be the rise in the temperature of the water? (b) Suppose the 6.0 Ω resistor gets burnt. What would be the rise in the temperature of the water in the next 15 minutes?
Concept: undefined >> undefined
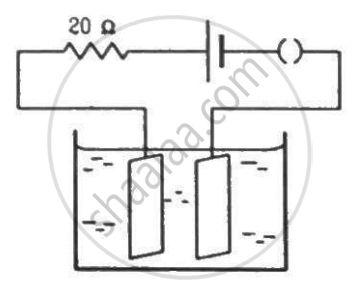
The figure shows an electrolyte of AgCl through which a current is passed. It is observed that 2.68 g of silver is deposited in 10 minutes on the cathode. Find the heat developed in the 20 Ω resistor during this period. Atomic weight of silver is 107.9 g/mol−1.
Concept: undefined >> undefined
A long, straight wire carries a current i. The magnetising field intensity H is measured at a point P close to the wire. A long, cylindrical iron rod is brought close to the wire, so that the point P is at the centre of the rod. The value of H at P will ______________ .
Concept: undefined >> undefined
Which of the following pairs has quantities of the same dimensions?
(a) Magnetic field B and magnetising field intensity H
(b) Magnetic field B and intensity of magnetisation I
(c) Magnetising field intensity H and intensity of magnetisation I
(d) Longitudinal strain and magnetic susceptibility
Concept: undefined >> undefined
Mark out the correct options.
(a) Diamagnetism occurs in all materials.
(b) Diamagnetism results from the partial alignment of permanent magnetic moment.
(c) The magnetising field intensity, H, is always zero in free space.
(d) The magnetic field of induced magnetic moment is opposite the applied field.
Concept: undefined >> undefined
The magnetic intensity H at the centre of a long solenoid carrying a current of 2.0 A, is found to be 1500 A m−1. Find the number of turns per centimetre of the solenoid.
Concept: undefined >> undefined
The magnetic field inside a long solenoid of 50 turns cm−1 is increased from 2.5 × 10−3 T to 2.5 T when an iron core of cross-sectional area 4 cm2 is inserted into it. Find (a) the current in the solenoid (b) the magnetisation I of the core and (c) the pole strength developed in the core.
Concept: undefined >> undefined
The magnetic field B and the magnetic intensity H in a material are found to be 1.6 T and 1000 A m−1, respectively. Calculate the relative permeability µr and the susceptibility χ of the material.
Concept: undefined >> undefined
Assume that each iron atom has a permanent magnetic moment equal to 2 Bohr magnetons (1 Bohr magneton equals 9.27 × 10−24 A m2). The density of atoms in iron is 8.52 × 1028 atoms m−3. (a) Find the maximum magnetisation I in a long cylinder of iron (b) Find the maximum magnetic field B on the axis inside the cylinder.
Concept: undefined >> undefined
Two circular loops are placed with their centres separated by a fixed distance. How would you orient the loops to have (a) the largest mutual inductance (b) the smallest mutual inductance?
Concept: undefined >> undefined
A rectangular coil of 100 turns has length 5 cm and width 4 cm. It is placed with its plane parallel to a uniform magnetic field and a current of 2 A is sent through the coil. Find the magnitude of the magnetic field B if the torque acting on the coil is 0.2 N m−1
Concept: undefined >> undefined
Is p − E/c valid for electrons?
Concept: undefined >> undefined
The frequency and intensity of a light source are doubled. Consider the following statements.
(A) The saturation photocurrent remains almost the same.
(B) The maximum kinetic energy of the photoelectrons is doubled.
Concept: undefined >> undefined
A non-monochromatic light is used in an experiment on photoelectric effect. The stopping potential
Concept: undefined >> undefined
