Please select a subject first
Advertisements
Advertisements
The standard reaction Gibbs energy for a chemical reaction at an absolute temperature T is given by ΔG0 = A − BT where A and Bare non-zero constants. Which of the following is true about this reaction?
Concept: undefined >> undefined
A diluted solution of sulphuric acid is electrolyzed using a current of 0.10 A for 2 hours to produce hydrogen and oxygen gas. The total volume of gases produced at STP is ______ cm3. (Nearest integer)
[Given: Faraday constant F = 96500 C mol–1 at STP, molar volume of an ideal gas is 22.7 L mol–1]
Concept: undefined >> undefined
Advertisements
The end product Y in the sequence of reaction:
\[\ce{RX ->[CN^-] X ->[NaOH] Y}\] is:
Concept: undefined >> undefined
A reddish-pink substance on heating gives off a vapour which condenses on the sides of the test tube and the substance turns blue. If on cooling water is added to the residue it turns to original colour. The substance is ______.
Concept: undefined >> undefined
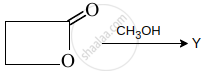
Y is:
Concept: undefined >> undefined
Alkaline hydrolysis of C4H8Cl2 gives a compound (A) which on heating with NaOH and I2 produces a yellow precipitate of CHI3. The compound (A) should be ______.
Concept: undefined >> undefined
Among the following which is the best description of water in the solid phase?
Concept: undefined >> undefined
A compound 'X' with molecular formula C3H8O can be oxidised to a compound 'Y' with the molecular formula C3H6O2 'X' is most likely to be ______.
Concept: undefined >> undefined
Hex-4-ene-2-ol on treatment with PCC gives 'A'. 'A' on reaction with sodium hypoiodite gives 'B', which on further heating with soda lime gives 'C. The compound 'C' is ______.
Concept: undefined >> undefined
How many unit cells are present in a cube-shaped ideal crystal of NaCl of mass 1.00 g? [Atomic masses: Na = 23, Cl = 35.5]
Concept: undefined >> undefined
Two statements are given below:
Statement I: The melting point of monocarboxylic acid with even number of carbon atoms is higher than that of with an odd number of carbon atoms acid immediately below and above it in the series.
Statement II: The solubility of monocarboxylic acids in water decreases with increased molar mass.
Choose the most appropriate option:
Concept: undefined >> undefined
Which one of the following acids does not exhibit optical isomerism?
Concept: undefined >> undefined
\[\ce{CH3CH2COOH ->[Cl2][red P] A ->[alc. KOH] B}\]. What is B?
Concept: undefined >> undefined
Orthorhombic crystal has the following unit cell dimensions ______.
Concept: undefined >> undefined
Assertion A: Enol form of acetone [CH3COCH3] exists in < 0.1% quantity. However, the enol form the acetyl acetone [CH3COCH2OCCH3] exists in approximately 15% quantity.
Reason R: Enol form of acetyl acetone is stabilized by intramolecular hydrogen bonding, which is not possible in enol form of acetone.
Choose the correct answer:
Concept: undefined >> undefined
A certain element crystallises in a bcc lattice of unit cell edge length 27 A°. If the same element under the same conditions crystallises in the fcc lattice, the edge length of the unit cell in A° will be ______. (Round off to the Nearest Integer).
[Assume each lattice point has a single atom]
Concept: undefined >> undefined
When propionamide reacts with Br2 in the presence of alkali the product is ______.
Concept: undefined >> undefined
Na2CO3 cannot be used in place of (NH4)2CO3 for the precipitation of the V group because ______.
Concept: undefined >> undefined
Gold crystallizes in a face-centered cubic lattice. If the length of the edge of the unit cell is 407 pm. The density of gold assuming it to be spherical is ______ g/cm3. Atomic mass of gold = 197 amu.
Concept: undefined >> undefined
The metallic luster exhibited by sodium is explained by ______.
Concept: undefined >> undefined
