Please select a subject first
Advertisements
Advertisements
\[\begin{array}{cc}
\ce{D}\phantom{........................}\\
|\phantom{.........................}\\
\ce{2D - C = O + OH^- ->[Cannizzaro] X and Y}
\end{array}\]
(Y is alcohol, D is deuterium)
X and Y will have the structure:
Concept: undefined >> undefined
In the Cannizzaro reaction given below:
\[\ce{2Ph-CHO ->[OH^-] Ph-CH2OH + PhC\overset{-}{O}_2}\]
the slowest step is:
Concept: undefined >> undefined
Advertisements
The first and second dissociation constants of an acid H2A are 1.0 × 10−5 and 5.0 × 10−10 respectively. The overall dissociation constant of the acid will be ______.
Concept: undefined >> undefined
Benzene on nitration gives nitrobenzene in presence of HNO3 and H2SO4 mixture, where ______.
Concept: undefined >> undefined
Which of the following would not give 2-phenylbutane as the major product in a Friedel-Crafts alkylation reaction?
Concept: undefined >> undefined
Complete combustion of 750 g of an organic compound provides 420 g of CO2 and 210 g of H2O. The percentage composition of carbon and hydrogen in organic compound is 15.3 and ______ respectively. (Round off to the Nearest Integer).
Concept: undefined >> undefined
These are physical properties of an elements.
- Sublimation enthalpy
- Ionisation enthalpy
- Hydration enthalpy
- Electron gain enthalpy
The total number of above properties that affect the reduction potential is ______. (Integer answer)
Concept: undefined >> undefined
What would be the product when neopentyl chloride reacts with sodium ethoxide?
Concept: undefined >> undefined
The relative order of reactivity of acyl derivatives is:
Concept: undefined >> undefined
The process with negative entropy change is ______.
Concept: undefined >> undefined
A reaction at 1 bar is non-spontaneous at low temperatures but becomes spontaneous at high temperatures. Identify the correct statement about the reaction among the following.
Concept: undefined >> undefined
Using the rules for significant figures, the correct answer for the expression `(0.02858 xx 0.112)/(0.5702)` will be ______.
Concept: undefined >> undefined
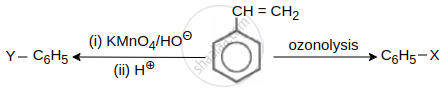
Functional groups Y, –CH = CH2 and X respectively are ______ and ______ directing.
Concept: undefined >> undefined
For the cell process:
\[\ce{Sn(s) + Pb^{+2}(aq) -> Pb(s) + Sn^{+2}(aq)}\]
The ratio of Pb+2 to Sn+2 ion concentration for spontaneity is ______.
Given: `"E"_("Sn"^(2+)//"Sn")^0` = − 0.136 V, `"E"_("Pb"^(2+)//"Pb")^0` = − 0.126 V
Concept: undefined >> undefined
The major aromatic product C in the following reaction sequence will be:
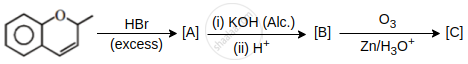
Concept: undefined >> undefined
For the reaction \[\ce{A(g) -> B(g)}\], the value of equilibrium constant at 300 K and 1 atm is equal to 100.0. The value of ΔrG for the reaction at 300 K and 1 atm in J mol−1 is −xR, where x is ______. (Rounded-off to the nearest integer)
[R = 8.31 J mol−1 K−1 and ln 10 = 2.3]
Concept: undefined >> undefined
Assertion A: Sharp glass edge becomes smooth on heating it upto its melting point.
Reason R: The viscosity of glass decreases on melting.
Choose the most appropriate answer from the options given below.
Concept: undefined >> undefined
An ionic solid AB2 isomorphous to the rutile structure (a tetragonal system with the effective number of formula units = 2) has edge lengths of the unit cell of 4Å, 4Å and 7Å. The density of the substance is ______ mg/cc. (if its formula weight is 80. The NA = 6 × 1023 and express your answer in mg/cc using four significant digits.)
Concept: undefined >> undefined
Ozone on reacting with KI in a neutral medium produces ______.
Concept: undefined >> undefined
To produce 1,4-dicyanobutane following reduction is carried out:
\[\ce{2CH2 = CH - CN + 2H+ + 2e- -> NC - (CH2)4) - CN}\]
The current ______ (in Ampere) must be used to produce 162 g of 1, 4-dicyanobutane per hour.
Concept: undefined >> undefined
