Please select a subject first
Advertisements
Advertisements

Which among the above compound/s does/do not form Silver mirror when treated with Tollen's reagent?
Concept: undefined >> undefined
Among the following, the aromatic compounds are:
| (A) | 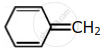 |
| (B) |  |
| (C) | 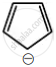 |
| (D) | 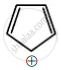 |
Choose the correct answer from the following options:
Concept: undefined >> undefined
Advertisements
The major products formed in the following reaction sequence A and B are:

Concept: undefined >> undefined
An organic compound neither reacts with neutral ferric chloride solution nor with Fehling solution. It however, reacts with Grignard reagent and gives positive iodoform test. The compound is:
Concept: undefined >> undefined
The correct set of products obtained in the following reactions:
- \[\ce{RCN ->[reduction]}\]
- \[\ce{RCN ->[(i) CH3MgBr][(ii) H2O]}\]
- \[\ce{RNC ->[hydrolysis]}\]
- \[\ce{RNH2 ->[HNO2]}\]
Concept: undefined >> undefined
Bonding in which of the following diatomic molecule(s) become(s) stronger, on the basis of MO Theory, by removal of an electron?
(A) NO
(B) N2
(C) O2
(D) C2
(E) B2
Choose the most appropriate answer from the options given below:
Concept: undefined >> undefined
The volume of gas A is twice than that of gas B. The compressibility factor of gas A is thrice than that of gas B at the same temperature. The pressures of the gases for an equal number of moles are ______.
Concept: undefined >> undefined
Choose the reaction which is not possible:
Concept: undefined >> undefined
In Tollen's test for aldehyde, the overall number of electrons(s) transferred to the Tollen's reagent formula \[\ce{[Ag(NH3)2]+}\] per aldehyde group to form silver mirror is ______. (Round off to the nearest integer)
Concept: undefined >> undefined
The oxoacid of phosphorus that is easily obtained from a reaction of alkali and white phosphorus and has two P-H bonds, is ______.
Concept: undefined >> undefined
Match List-I with List-II:
| List-I | List-II |
| name of oxo acid | Oxidation state of 'P' |
| (a) Hypophosphorous acid | (i) +5 |
| (b) Orthophosphoric acid | (ii) +4 |
| (c) Hypophosphoric acid | (iii) +3 |
| (d) Orthophosphorous acid | (iv) +2 |
| (v) +1 |
Choose the correct answer from the options given below:
Concept: undefined >> undefined
\[\ce{CH3-CH2-CHO ->[dil][alkali] Product}\]
The product in the above reaction is:
Concept: undefined >> undefined
Benzene reacts with benzoyl chloride to form ______.
Concept: undefined >> undefined
Phenol associates in benzene to a certain extent to form a dimer. A solution containing 20 × 10-3 kg of phenol in 1.0 kg of benzene has its freezing point depressed by 0.69 K The fraction of phenol that has dimerised is ______. (Kf for benzene = 5.12 K kg mol-1)
Concept: undefined >> undefined
The major product of the following reaction is:

Concept: undefined >> undefined
In van der Waals equation of state of the gas law, the constant 'b' is a measure of ______.
Concept: undefined >> undefined
In Friedel-Crafts alkylation of aniline, one gets ______.
Concept: undefined >> undefined
Excess of isobutane on reactions with Br2 in presence of light at 125°C gives which one of the following, as the major product?
Concept: undefined >> undefined
These are physical properties of an elements.
- Sublimation enthalpy
- Ionisation enthalpy
- Hydration enthalpy
- Electron gain enthalpy
The total number of above properties that affect the reduction potential is ______. (Integer answer)
Concept: undefined >> undefined
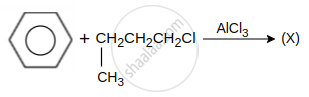
hydrocarbon (X) major product X is:
Concept: undefined >> undefined
