Please select a subject first
Advertisements
Advertisements
Formic acid and formaldehyde can be distinguished by treating with ______.
Concept: undefined >> undefined
Which primitive unit cell has unequal edge lengths (a ≠ b ≠ c) and all axial angles different from 90°?
Concept: undefined >> undefined
Advertisements
Hydrogen ion concentration in mol/L in a solution of pH = 5.4 will be ______.
Concept: undefined >> undefined
Assuming that the degree of hydrolysis is small, the pH of 0.1 M solution of sodium acetate (Ka = 1.0 × 10−5) will be ______.
Concept: undefined >> undefined
The pH of a 0.02 M NH4Cl solution will be ______.
[Given: Kb(NH4OH] = 10−5 and log 2 = 0.301)
Concept: undefined >> undefined
Given below are two statements:
Statement I: The esterification of carboxylic acid with an alcohol is a nucleophilic acyl substitution.
Statement II: Electron withdrawing groups in the carboxylic acid will increase the rate of esterification reactions.
Choose the most appropriate options.
Concept: undefined >> undefined
In the given reaction,

The number of n electrons present in the product 'P' is ______.
Concept: undefined >> undefined
In an ionic solid r(+) = 1.6 Å and r(−) = 1.864 Å. Use the radius ratio rule to the edge length of the cubic unit cell is ______ Å.
Concept: undefined >> undefined
An element A (Atomic weight = 100) having bcc structure has a unit cell edge length 400 pm. The number of atoms in 10 g of A is ______ × 1022 unit cells.
Concept: undefined >> undefined
A reaction of ethyl amine and acetic anhydride leads to the formation of ______.
Concept: undefined >> undefined
The ratio of number of atoms present in a simple cubic, body-centred cubic and face-centred cubic structure are, respectively ______.
Concept: undefined >> undefined
Which of the following oxoacids of sulphur contains "S" in two different oxidation states?
Concept: undefined >> undefined
The solubility of AgCl will be maximum in which of the following?
Concept: undefined >> undefined
The incorrect statements is:
Concept: undefined >> undefined
For the following Assertion and Reason, the correct option is:
Assertion (A): When Cu (II) and sulphide ions are mixed they react together extremely quickly to give a solid.
Reason (R): The equilibrium constant of \[\ce{Cu^{2+}(aq) + S^{2-}(aq) <=> CuS(s)}\] is high because the solubility product is low.
Concept: undefined >> undefined
An organic compound X showing the following solubility profile is:
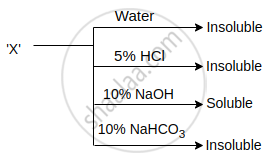
Concept: undefined >> undefined
If solubility product of Zr3(PO4)4 is denoted by Ksp and its molar solubility is denoted by S, then which of the following relation between S and Ksp is correct?
Concept: undefined >> undefined
Identify product A in the following reaction.
\[\begin{array}{cc}
\ce{COOH}\phantom{.......}\\
/\phantom{...............}\\
\ce{CH2 ->[\Delta] CH3COOH + A}\\
\backslash\phantom{................}\\
\ce{COOH}\phantom{........}
\end{array}\]
Concept: undefined >> undefined
Water soluble salt among AgNO3, AgF and AgClO4 are:
Concept: undefined >> undefined
Which of the following salts will turn water coloured when fumes evolved on treatment with conc. H2SO4 are passed in water?
Concept: undefined >> undefined
