Please select a subject first
Advertisements
Advertisements
Differentiate between the following pairs based on the odourless gas which turns lime water milky and the criteria given:
Sulphuric acid and Nitric acid (using barium chloride solution)
Concept: undefined >> undefined
Arrange the following as per the instruction given in the bracket:
Sulphuric acid, Phosphoric acid, Acetic acid (increasing order of number of replaceable H atoms per molecule).
Concept: undefined >> undefined
Advertisements
Identify the following:
The element having electronic configuration 2, 8, 6.
Concept: undefined >> undefined
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
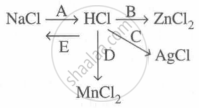
Concept: undefined >> undefined
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
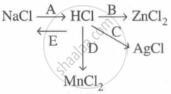
Concept: undefined >> undefined
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
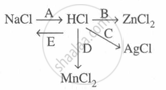
Concept: undefined >> undefined
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCI->}\]
Concept: undefined >> undefined
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl ->}\]
Concept: undefined >> undefined
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NH4OH + HCl->}\]
Concept: undefined >> undefined
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
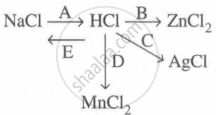
Concept: undefined >> undefined
1H2 is an isotope of hydrogen. In the modern Periodic Table it will ______.
Concept: undefined >> undefined
Identify the following:
A homogenous mixture of two or more metals or a metal and a non-metal in a definite proportion in their molten state.
Concept: undefined >> undefined
Draw the structural diagram for the following compound.
propanoic acid
Concept: undefined >> undefined
What property of Sulphuric acid is exhibited in the following case?
In the preparation of HCI gas when it reacts with Sodium chloride.
Concept: undefined >> undefined
You are provided with the list of chemicals mentioned below in the box:
| Sodium hydroxide solution, copper carbonate, zinc, hydrochloric acid, copper, dilute sulphuric acid, chlorine, iron |
Using suitable chemicals from the list given, write a balanced chemical equation for the preparation of the salt mentioned below:
Copper sulphate
Concept: undefined >> undefined
You are provided with the list of chemicals mentioned below in the box:
| Sodium hydroxide solution, copper carbonate, zinc, hydrochloric acid, copper, dilute sulphuric acid, chlorine, iron |
Using suitable chemicals from the list given, write a balanced chemical equation for the preparation of the salt mentioned below:
Sodium zincate
Concept: undefined >> undefined
You are provided with the list of chemicals mentioned below in the box:
| Sodium hydroxide solution, copper carbonate, zinc, hydrochloric acid, copper, dilute sulphuric acid, chlorine, iron |
Using suitable chemicals from the list given, write a balanced chemical equation for the preparation of the salt mentioned below:
Ferric chloride
Concept: undefined >> undefined
Give a balanced equation for the following:
Oxidation of carbon with conc. nitric acid.
Concept: undefined >> undefined
Name the alloy which is made up of Copper, Zinc and Tin.
Concept: undefined >> undefined
| Abhishek was given a salt ‘X’ which was white in colour for analysis. On strong heating, it produced a yellow residue, a colourless gas and also a reddish-brown gas. The solution of the salt ‘X’, when tested with an excess of ammonium hydroxide, produced a chalky white insoluble precipitate. |
- Name the coloured gas that evolved when Abhishek heated the salt strongly.
- Which cation was present in the sample given to Abhishek?
- Identify the salt given to Abhishek for analysis.
Concept: undefined >> undefined
