Please select a subject first
Advertisements
Advertisements
X [2, 8, 7] and Y [2, 8, 2] are two elements. Using this information, complete the following:
______ is the metallic element.
Concept: undefined >> undefined
X [2, 8, 7] and Y [2, 8, 2] are two elements. Using this information, complete the following:
Metal atoms tend to have a maximum of ______ electrons in the outermost shell.
Concept: undefined >> undefined
Advertisements
X [2, 8, 7] and Y [2, 8, 2] are two elements. Using this information, complete the following:
______ is the reducing agent.
Concept: undefined >> undefined
State the group and period, of the element having three shells with three electrons in valence shell.
Concept: undefined >> undefined
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
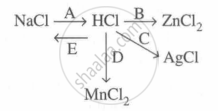
Concept: undefined >> undefined
Name two elements that you would expect to show chemical reactions similar to calcium. What is the basis of your choice?
Concept: undefined >> undefined
Name the metalloids in the first twenty elements.
Concept: undefined >> undefined
Name the non-metals in the first twenty elements.
Concept: undefined >> undefined
What is the general name of the last elements of the Periodic Table?
Concept: undefined >> undefined
Name the following metal:
A metal present in period 3, group 1 of the periodic table.
Concept: undefined >> undefined
An element has 2 electrons in its N shell.
State its position in periodic table.
Concept: undefined >> undefined
An element has 2 electrons in its N shell.
Is it a metal or a non-metal?
Concept: undefined >> undefined
Answer the following in respect of element \[\ce{_16^32S}\].
Give its electronic configuration.
Concept: undefined >> undefined
Answer the following in respect of element \[\ce{_16^32S}\].
To which group and period does it belong?
Concept: undefined >> undefined
Answer the following in respect of element \[\ce{_16^32S}\].
Give its formula with hydrogen.
Concept: undefined >> undefined
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
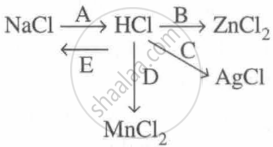
Concept: undefined >> undefined
Match the atomic number with the following:
A rare gas.
Concept: undefined >> undefined
Match the atomic number with the following:
An element of group 2.
Concept: undefined >> undefined
Give reason for the following:
Inert gases do not form ion.
Concept: undefined >> undefined
A group of elements in the periodic table is given below (Boron is the first member of the group and Thallium is the last).
Boron, Aluminium, Gallium, Indium, Thallium
Answer the following question in relation to the above group of elements:
If the electronic configuration of aluminium is 2, 8, 3, how many electrons are there in the outer shell of thallium?
Concept: undefined >> undefined
