Advertisements
Advertisements
Assume that there is no repulsive force between the electrons in an atom but the force between positive and negative charges is given by Coulomb’s law as usual. Under such circumstances, calculate the ground state energy of a He-atom.
Concept: undefined >> undefined
The Bohr model for the H-atom relies on the Coulomb’s law of electrostatics. Coulomb’s law has not directly been verified for very short distances of the order of angstroms. Supposing Coulomb’s law between two opposite charge + q1, –q2 is modified to |F| = `(q_1q_2)/((4πε_0)) 1/r^2, r ≥ R_0 = (q_1q_2)/(4πε_0) 1/R_0^2 (R_0/r)^ε, r ≤ R_0` Calculate in such a case, the ground state energy of a H-atom, if ε = 0.1, R0 = 1Å.
Concept: undefined >> undefined
Advertisements
Explain why elemental semiconductor cannot be used to make visible LEDs.
Concept: undefined >> undefined
The frequency response curve (Figure) for the filter circuit used for production of AM wave should be ______.
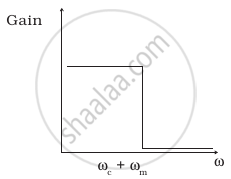 (i) |
 (ii) |
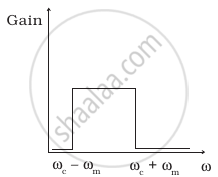 (iii) |
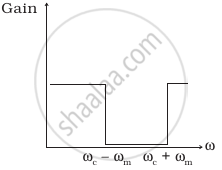 (iv) |
- (i) followed by (ii).
- (ii) followed by (i).
- (iii).
- (iv).
Concept: undefined >> undefined
The electric potential on the axis of an electric dipole at a distance ‘r from it’s centre is V. Then the potential at a point at the same distance on its equatorial line will be ______.
Concept: undefined >> undefined
A long straight wire of circular cross section of radius 'a' carries a steady current I. The current is uniformly distributed across its cross section. The ratio of magnitudes of the magnetic field at a point `a/2` above the surface of wire to that of a point `a/2` below its surface is ______.
Concept: undefined >> undefined
If the reading of the voltmeter V1 is 40 V, then the reading of voltmeter V2 is ______.

Concept: undefined >> undefined
ASSERTION (A): The electrical conductivity of a semiconductor increases on doping.
REASON (R): Doping always increases the number of electrons in the semiconductor.
Concept: undefined >> undefined
Electromagnetic waves with wavelength
- λ1 is suitable for radar systems used in aircraft navigation.
- λ2 is used to kill germs in water purifiers.
- λ3 is used to improve visibility in runways during fog and mist conditions.
Identify and name the part of the electromagnetic spectrum to which these radiations belong. Also arrange these wavelengths in ascending order of their magnitude.
Concept: undefined >> undefined
What is the nuclear radius of 125Fe, if that of 27Al is 3.6 fermi?
Concept: undefined >> undefined
Draw a graph showing the variation of the number of particles scattered (N) with the scattering angle θ in the Geiger-Marsden experiment. Why only a small fraction of the particles are scattered at θ > 90°?
Concept: undefined >> undefined
Arrange the following electromagnetic radiation in the ascending order of their frequencies:
X-rays, microwaves, gamma rays, radio waves
Concept: undefined >> undefined
Write two uses of the following radiation.
X-rays
Concept: undefined >> undefined
Write two uses of the following radiation.
Gamma rays
Concept: undefined >> undefined
Give an example each of a metal from which photoelectric emission takes place when irradiated by
- UV light
- visible light.
Concept: undefined >> undefined
Name the factors on which photoelectric emission from a surface depends.
Concept: undefined >> undefined
A narrow beam of protons, each having 4.1 MeV energy is approaching a sheet of lead (Z = 82). Calculate:
- the speed of a proton in the beam, and
- the distance of its closest approach
Concept: undefined >> undefined
Electromagnetic waves of wavelengths λ1, λ2 and λ3 are used in a radar system, in water purifiers and in remote switches of TV, respectively.
- Identify the electromagnetic waves.
- Write one source for each of them.
Concept: undefined >> undefined
The work function of a metal is 2.31 eV. Photoelectric emission occurs when the light of frequency 6.4 × 1014 Hz is incident on the metal surface. Calculate
- the energy of the incident radiation,
- the maximum kinetic energy of the emitted electron and
- the stopping potential of the surface.
Concept: undefined >> undefined
The energy of hydrogen atom in an orbit is −1.51 eV. What are the kinetic and potential energies of the electron in this orbit?
Concept: undefined >> undefined
