Advertisements
Advertisements
Discuss biological and industrial importance of osmosis.
Concept: undefined >> undefined
How can you remove the hard calcium carbonate layer of the egg without damaging its semiprermiable membrane? Can this egg be inserted into a bottle with a narrow neck without distorting its shape? Explain the process involved.
Concept: undefined >> undefined
Advertisements
At high concentration of soap in water, soap behaves as ______.
Concept: undefined >> undefined
Which of the following options are correct?
(i) Micelle formation by soap in aqueous solution is possible at all temperatures.
(ii) Micelle formation by soap in aqueous solution occurs above a particular concentration.
(iii) On dilution of soap solution micelles may revert to individual ions.
(iv) Soap solution behaves as a normal strong electrolyte at all concentrations.
Concept: undefined >> undefined
Method of formation of solution is given in Column I. Match it with the type of solution given in Column II.
| Column I | Column II |
| (i) Sulphur vapours passed through cold water | (a) Normal electrolyte solution |
| (ii) Soap mixed with water above critical micelle concentration | (b) Molecular colloids |
| (iii) White of egg whipped with water | (c) Associated colloid |
| (iv) Soap mixed with water below critical micelle concentration | (d) Macro molecular colloids |
Concept: undefined >> undefined
A chelating agent has two or more than two donor atoms to bind to a single metal ion. Which of the following is not a chelating agent?
Concept: undefined >> undefined
Which of the following species is not expected to be a ligand?
Concept: undefined >> undefined
Assertion: Toxic metal ions are removed by the chelating ligands.
Reason: Chelate complexes tend to be more stable.
Concept: undefined >> undefined
Molecules whose mirror image is non-superimposable over them are known as chiral. Which of the following molecules is chiral in nature?
Concept: undefined >> undefined
Which of the following statements are correct about the mechanism of this reaction?
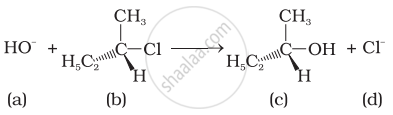
(i) A carbocation will be formed as an intermediate in the reaction.
(ii) \[\ce{OH-}\] will attach the substrate (b) from one side and \[\ce{Cl-}\] will leave it simultaneously from other side.
(iii) An unstable intermediate will be formed in which \[\ce{OH-}\] and \[\ce{Cl-}\] will be attached by weak bonds.
(iv) Reaction proceeds through SN1 mechanism.
Concept: undefined >> undefined
The correct order of increasing acidic strength is ______.
Concept: undefined >> undefined
Why is \[\ce{NH2}\] group of aniline acetylated before carrying out nitration?
Concept: undefined >> undefined
Coagulation of egg white on boiling is an example of denaturation of protein. Explain it in terms of structural changes.
Concept: undefined >> undefined
Complete the following:
\[\ce{C6H5NO2 ->[Sn/HCl] A ->[Br2/H2O] B ->[NaNO2/HCl][273 - 278 K] C ->[HBF4][Δ] D}\]
Concept: undefined >> undefined
What would be the major product of the following reaction?
\[\ce{C6H5 - CH2 - OC6H5 + HBr -> A + B}\]
Concept: undefined >> undefined
Differentiate between the following:
Multimolecular Colloid and Macromolecular colloid
Concept: undefined >> undefined
Assertion (A) : Osmotic pressure is a colligative property.
Reason (R) : Osmotic pressure is proportional to the molality.
Concept: undefined >> undefined
Determine the osmotic pressure of a solution prepared by dissolving 2.32 × 10−2 g of K2SO4 in 2L of solution at 25°C assuming that K2SO4 is completely dissociated.
(R = 0.082 L atm K−1 mol, Molar mass K2SO4 = 174 g mol−1)
Concept: undefined >> undefined
What structural change will occur when a native protein is subjected to change in pH?
Concept: undefined >> undefined
What is meant by the chelate effect? Give an example.
Concept: undefined >> undefined
