Please select a subject first
Advertisements
Advertisements
Do two distinct poles actually exist at two nearby points in a magnetic dipole?
Concept: undefined >> undefined
A circular loop carrying a current is replaced by an equivalent magnetic dipole. A point on the axis of the loop is in
Concept: undefined >> undefined
Advertisements
A circular loop carrying a current is replaced by an equivalent magnetic dipole. A point on the loop is in ______.
Concept: undefined >> undefined
When a current in a circular loop is equivalently replaced by a magnetic dipole,
Concept: undefined >> undefined
Let r be the distance of a point on the axis of a bar magnet from its centre. The magnetic field at such a point is proportional to
Concept: undefined >> undefined
Let r be the distance of a point on the axis of a magnetic dipole from its centre. The magnetic field at such a point is proportional to
Concept: undefined >> undefined
Two short magnets of equal dipole moments M are fastened perpendicularly at their centre in the Figure . The magnitude of the magnetic field at a distance d from the centre on the bisector of the right angle is
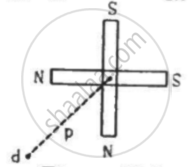
Concept: undefined >> undefined
Pick the correct options.
(a) Magnetic field is produced by electric charges only
(b) Magnetic poles are only mathematical assumptions having no real existence
(b) A north pole is equivalent to a clockwise current and a south pole is equivalent to an anticlockwise current.
(d) A bar magnet is equivalent to a long, straight current.
Concept: undefined >> undefined
A horizontal circular loop carries a current that looks clockwise when viewed from above. It is replaced by an equivalent magnetic dipole consisting of a south pole S and a north pole N.
(a) The line SN should be along a diameter of the loop.
(b) The line SN should be perpendicular to the plane of the loop
(c) The south pole should be slow the loop
(d) The north pole should be below the loop
Concept: undefined >> undefined
A magnetic dipole of magnetic moment 0.72 A m2 is placed horizontally with the north pole pointing towards south. Find the position of the neutral point if the horizontal component of the earth's magnetic field is 18 μT.
Concept: undefined >> undefined
In a radioactive decay, neither the atomic number nor the mass number changes. Which of the following particles is emitted in the decay?
Concept: undefined >> undefined
A freshly prepared radioactive source of half-life 2 h emits radiation of intensity which is 64 times the permissible safe level. The minimum time after which it would be possible to work safely with this source is
Concept: undefined >> undefined
The decay constant of a radioactive sample is λ. The half-life and the average-life of the sample are respectively
Concept: undefined >> undefined
Lithium (Z = 3) has two stable isotopes 6Li and 7Li. When neutrons are bombarded on lithium sample, electrons and α-particles are ejected. Write down the nuclear process taking place.
Concept: undefined >> undefined
The masses of 11C and 11B are respectively 11.0114 u and 11.0093 u. Find the maximum energy a positron can have in the β*-decay of 11C to 11B.
(Use Mass of proton mp = 1.007276 u, Mass of `""_1^1"H"` atom = 1.007825 u, Mass of neutron mn = 1.008665 u, Mass of electron = 0.0005486 u ≈ 511 keV/c2,1 u = 931 MeV/c2.)
Concept: undefined >> undefined
28Th emits an alpha particle to reduce to 224Ra. Calculate the kinetic energy of the alpha particle emitted in the following decay:
`""^228"Th" → ""^224"Ra"^(∗) + alpha`
`""^224"Ra"^(∗) → ""^224"Ra" + γ (217 "keV")`.
Atomic mass of 228Th is 228.028726 u, that of 224Ra is 224.020196 u and that of `""_2^4H` is 4.00260 u.
(Use Mass of proton mp = 1.007276 u, Mass of `""_1^1"H"` atom = 1.007825 u, Mass of neutron mn = 1.008665 u, Mass of electron = 0.0005486 u ≈ 511 keV/c2,1 u = 931 MeV/c2.)
Concept: undefined >> undefined
Calculate the maximum kinetic energy of the beta particle emitted in the following decay scheme:
12N → 12C* + e+ + v
12C* → 12C + γ (4.43MeV).
The atomic mass of 12N is 12.018613 u.
(Use Mass of proton mp = 1.007276 u, Mass of `""_1^1"H"` atom = 1.007825 u, Mass of neutron mn = 1.008665 u, Mass of electron = 0.0005486 u ≈ 511 keV/c2,1 u = 931 MeV/c2.)
Concept: undefined >> undefined
The decay constant of `""_80^197`Hg (electron capture to `""_79^197`Au) is 1.8 × 10−4 S−1. (a) What is the half-life? (b) What is the average-life? (c) How much time will it take to convert 25% of this isotope of mercury into gold?
Concept: undefined >> undefined
The decay constant of 238U is 4.9 × 10−18 S−1. (a) What is the average-life of 238U? (b) What is the half-life of 238U? (c) By what factor does the activity of a 238U sample decrease in 9 × 109 years?
Concept: undefined >> undefined
57Co decays to 57Fe by β+- emission. The resulting 57Fe is in its excited state and comes to the ground state by emitting γ-rays. The half-life of β+- decay is 270 days and that of the γ-emissions is 10−8 s. A sample of 57Co gives 5.0 × 109 gamma rays per second. How much time will elapse before the emission rate of gamma rays drops to 2.5 × 109per second?
Concept: undefined >> undefined
