Definitions [9]
Define the term Hydrogen bond
The electrostatic force of attraction between a positively polarised hydrogen atom of one molecule and a highly electronegative atom (which may be negatively charged) of another molecule is called a hydrogen bond.
Define the term Dipole moment
Dipole moment (μ) is the product of the magnitude of the charge (Q) and the distance between the centres of positive and negative charge (r). It is designated by a Greek Letter (μ) and its unit is Debye (D).
Define the term polarizability
Polarizability is defined as the ability of an atom or a molecule to form momentary dipoles, which means, the ability of the atom or molecule to become polar by redistributing its electrons.
Define the term Aqueous tension
The pressure exerted by saturated water vapour is called aqueous tension.
Avogadro s law states that "equal volumes of all gases under similar conditions of temperature and pressure contain the same number of molecules."
An atom is the smallest particle of an element that can take part in a chemical reaction; however, it may or may not exist independently.
A molecule is the smallest particle of an element or a compound that can exist by itself; it never breaks up except for taking part in a chemical reaction.
Define interconversion of states of matter.
The process by which matter changes from one state to another and back to the original state, without any change in its chemical composition.
Define matter
Anything that has mass and occupies space is called matter.
Theorems and Laws [3]
Boyle's Law (Pressure–Volume Relationship)
At constant temperature (T) and number of moles (n), the pressure of a gas is inversely proportional to its volume.
\[P\propto\frac{1}{V}\quad\Rightarrow\quad P_1V_1=P_2V_2\]
The p–V curve at constant temperature is called an isotherm.
or
Statement:
For a fixed mass of gas at constant temperature, the pressure is inversely proportional to the volume.
Mathematically, P ∝ \[\frac {1}{V}\] ⇒ PV = constant
Graph: P vs V (Isotherm)
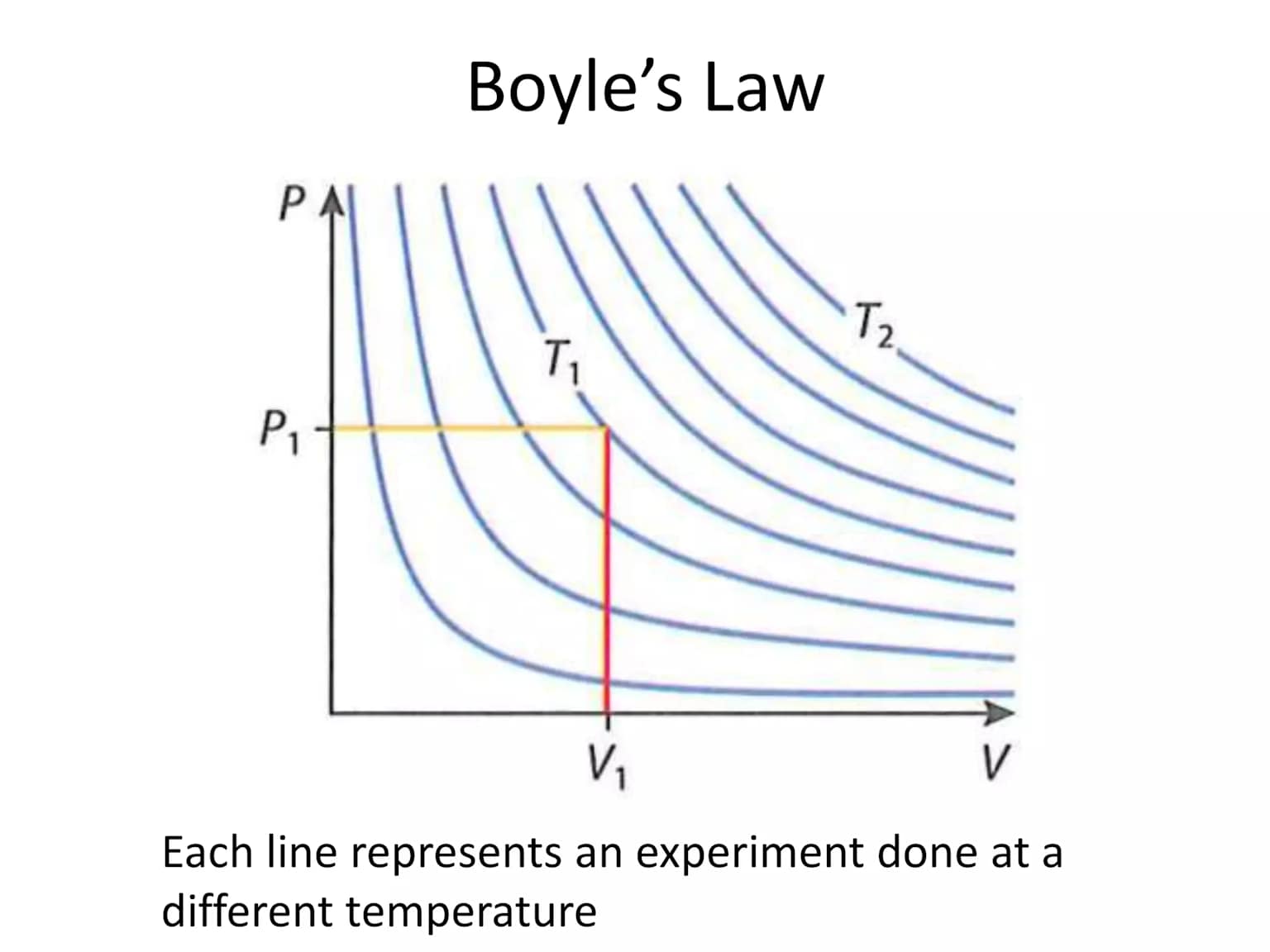
This means: squeezing a gas into a smaller space increases its pressure. Doubling the pressure halves the volume.
Charles' Law (Temperature–Volume Relationship)
At constant pressure (P) and number of moles (n), the volume of a gas is directly proportional to its absolute temperature.
\[V\propto T\quad\Rightarrow\quad\frac{V_1}{T_1}=\frac{V_2}{T_2}\]
The V–T curve at constant pressure is called an isobar
Absolute zero = 0 K = –273.15°C — the temperature at which gas volume theoretically becomes zero. It cannot be attained in practice (temperatures of ~0.000001 K have been achieved in labs)
or
Statement:
The volume of a fixed mass of gas is directly proportional to its absolute temperature if the pressure is kept constant.
Mathematically, V ∝ T ⇒ \[\frac {V}{T}\] = constant
Graph: V vs T (Isobar)
 A straight line through the origin when using Kelvin. All lines converge at 0 K (absolute zero).
A straight line through the origin when using Kelvin. All lines converge at 0 K (absolute zero).
Avogadro's Law (Volume–Moles Relationship):
At constant temperature (T) and pressure (P), volume is directly proportional to number of moles.
Key Points
Intermolecular forces are attractive (and repulsive) forces acting between neighbouring molecules. They are weaker than covalent or ionic bonds but determine the physical state of matter.
As intermolecular forces increase: Gas → Liquid → Solid (thermal energy decreases in the same direction).
Types of Intermolecular Forces:
| Type | Occurrence | Strength | Key Point | Example |
|---|---|---|---|---|
| Dipole–Dipole | Between polar molecules | Medium (3–4 kJ mol⁻¹) | +ve end attracts –ve end | HCl |
| Ion–Dipole | Between ion & polar molecule | Stronger than dipole–dipole | Depends on charge & size of ion | Na⁺ – H₂O |
| Dipole–Induced Dipole | Polar + non-polar molecule | Weak | Polar molecule induces dipole | NH₃ + C₆H₆ |
| London Dispersion | Non-polar molecules, noble gases | Weakest | Due to temporary dipoles | N₂, O₂, noble gases |
| Hydrogen Bonding | H with N, O, F | Strong (but < covalent) | Special dipole–dipole | HF, H₂O |
- Working Principle: Thermal power stations use steam to rotate turbines. Steam is generated by heating water using thermal energy from burning coal.
- Energy Conversion: Chemical energy in coal → Thermal energy → Kinetic energy in steam → Kinetic energy in turbine → Electrical energy via generator.
- Steam Recirculation: After rotating the turbine, steam is cooled in a condenser and converted back to water, which is then re-circulated to the boiler.
- Environmental Impact: Burning coal releases harmful gases (CO₂, SOx, NOx) and soot, causing air pollution and respiratory issues.
- Limitations: Coal reserves are limited, and dependence on them for electricity poses long-term sustainability issues.
- A gas consists of an extremely large number of tiny, discrete molecules whose actual volume is negligible compared to the total volume of the gas
- Gas molecules are in constant, random motion moving in straight lines; they change direction upon collisions with other molecules or container walls
- Intermolecular forces are negligible — molecules neither attract nor repel each other
- Effect of gravity on molecules is negligible
- All molecular collisions are perfectly elastic — total kinetic energy is conserved (though energy can be redistributed)
- Gas pressure is caused by molecular bombardment against the walls of the container
- Different molecules have different kinetic energies, but the average KE is directly proportional to absolute temperature: Average KE ∝ T
| Property | Solid | Liquid | Gas |
|---|---|---|---|
| Mean molecular separation | ~3–5 Å | ~3–10 Å | >5 Å |
| Particle arrangement | Tightly packed, regular | Loosely packed, irregular | Highly irregular |
| Particle movement | Fixed positions, cannot move freely | Moves a small distance within liquid | Continuous random motion |
| Shape & volume | Definite shape and volume | Takes shape of container, definite volume | Takes shape and volume of container |
| Intermolecular space | Very small | Moderate | Large |
| Effect of temperature | Small volume change | Moderate volume change | Significant volume change |
| Compressibility | Practically incompressible | Slightly compressible | Highly compressible |
| Example | A piece of iron | Water, spirit, oil | Air |
Concepts [21]
- Intermolecular Forces
- Dispersion Forces Or London Forces
- Dipole - Dipole Forces
- Dipole-induced Dipole Forces
- Hydrogen Bond
- Power Plant Based on Thermal Energy
- Intermolecular Forces Vs. Thermal Interactions
- The Gaseous State
- Boyle’s Law (Pressure - Volume Relationship)
- Charles’ Law (Temperature - Volume Relationship)
- Gay Lussac’s Law (Pressure- Temperature Relationship)
- Avogadro's Law
- Density and Molar Mass of a Gaseous Substance
- Dalton’s Law of Partial Pressures
- Kinetic Molecular Theory of Gases
- Behaviour of Real Gases: Deviation from Ideal Gas Behaviour
- Liquefaction of Gases
- Vapour Pressure
- Surface Tension
- Viscosity
- States of Matter
