Definitions [33]
Define the term solution.
A homogeneous mixture of two or more substances, whose relative amounts may be changed within certain limits, is called a solution.
A solution in which no solute can be dissolved further at a given temperature is called a saturated solution.
A solution which contains more solute than would be necessary to saturate it at a given temperature is called a supersaturated solution.
A solution in which more solute can be dissolved without raising the temperature is called an unsaturated solution.
Define isotonic solutions.
Two or more solutions exerting the same osmotic pressure are called isotonic solutions.
When two solutions are separated by a semipermeable membrane and no osmosis occurs, i.e., there is no net flow of water on either side through the membrane, the solutions are said to be isotonic solutions. If the membrane is perfectly semipermeable, the two solutions possess the same osmotic pressure and are also referred to as iso-osmotic solutions.
Define the term:
Ideal solution
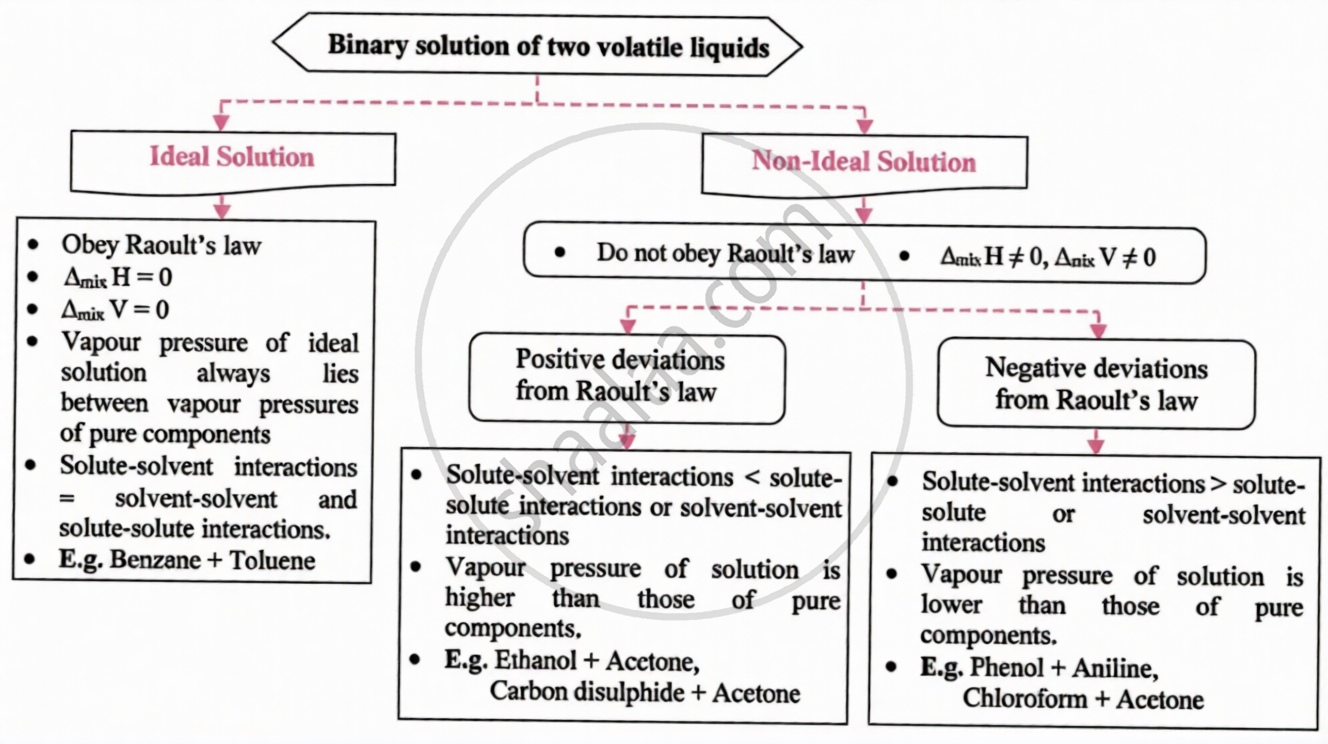
It is characterised as a solution that adheres to Raoult's Law, with no interactions between the molecules and no volume or heat change during mixing.
For an ideal solution, Enthalpy of mixing of the pure components to form the solution is Δmix H = 0 and the volume of mixing is Δmix V = 0.
It is defined as the amount of solute that can be dissolved in 100 g of the solvent at the given conditions. It is also expressed as the maximum quantity of solute moles that can be dissolved in solvent to form 1 dm³ of solution.
Define ebullioscopic constant.
Ebullioscopic constant or molal elevation constant, is the elevation in the boiling point produced when one mole of the solute is dissolved in one kilogram of solvent.
ΔTb = Kb × m
ΔTb = Kb if m = 1 molal
Where Kb = Molal elevation constant
ΔTb = Elevation in boiling point.
It is the net spontaneous flow of solvent molecules into the solution or from a more dilute solution to a more concentrated solution through a semipermeable membrane.
Osmotic pressure is the minimum pressure which needs to be applied to a solution to prevent the inward flow of its pure solvent across a semipermeable membrane.
\[\pi=\frac{n_2RT}{V}=\mathrm{CRT}\]
\[\pi=\frac{w_2RT}{\mathrm{M}_2V}\]
If a pressure larger than the osmotic pressure is applied to the solution side, then pure solvent from the solution passes into the pure solvent side through the semipermeable membrane. This phenomenon is called reverse osmosis.
or
Osmosis is a flow of solvent through a semipermeable membrane into the solution. The direction of osmosis can be reversed by applying a pressure larger than the osmotic pressure. This is called reverse osmosis.
Define Osmosis.
The net spontaneous flow of solvent molecules into the solution or from more dilute solution to more concentrated solution through a semipermeable membrane is called osmosis.
Define the following term:
Hypotonic solution
The solution having lower osmotic pressure as compared to some other solution is referred to as a hypotonic solution.
Define osmotic pressure.
Osmotic pressure may be defined as the external pressure which should be applied to the solution in order to stop the phenomenon of osmosis, i.e., to stop the flow of solvent into the solution when the two are separated by a semipermeable membrane.
Define Semipermeable membrane
Semipermeable membrane: It is a membrane which allows the solvent molecules, but not the solute molecules, to pass through it.
Semipermeable membrane is a film such as cellophane which has pores large enough to allow the solvent molecules to pass through them.
Define the following term:
isotonic solution
Two or more solutions exerting the same osmotic pressure are called an isotonic solution.
Define reverse osmosis.
The process of moving a solvent from a solution to a pure solvent through a semipermeable membrane while applying excessive pressure on the solution side is known as reverse osmosis.
It is a thin film, such as cellophane, which has pores large enough to allow the solvent molecules to pass through them.
or
When a solution and pure solvent or two solutions of different concentrations are separated by a semipermeable membrane, the solvent molecules pass through the membrane this is called osmosis.
A homogeneous mixture of two or more chemically non-reacting substances is called a solution.
The component of a solution which is present in smaller quantity and gets dissolved is called solute.
The component of a solution which is present in larger quantity and dissolves the solute is called solvent.
The maximum amount of solute that can be dissolved in a given amount of solvent at a specific temperature is called solubility.
A solution that contains the maximum amount of solute at a given temperature is called a saturated solution.
A solution that contains less solute than the maximum amount that can be dissolved at a given temperature is called an unsaturated solution.
A solution that contains more solute than required for saturation at a given temperature is called a supersaturated solution.
The pressure required to stop the flow of solvent through a semipermeable membrane during osmosis is called osmotic pressure.
The flow of solvent molecules through a semipermeable membrane from pure solvent or dilute solution to concentrated solution is called osmosis.
Two solutions having the same osmotic pressure at a given temperature are called isotonic solutions.
A solution having higher osmotic pressure than another solution is called a hypertonic solution.
A solution having lower osmotic pressure than another solution is called a hypotonic solution.
The physical properties of dilute solutions that depend only on the number of solute particles and not on their nature are called colligative properties.
The process in which solvent flows from solution into pure solvent through a semipermeable membrane by applying pressure greater than osmotic pressure is called reverse osmosis.
The fraction of total number of molecules that dissociate into ions is called degree of dissociation.
Formulae [1]
\[i=\frac{\text{Colligative property of electrolyte solution}}{\text{Colligative property of nonelectrolyte solution}}\]
\[i=\frac{\text{Actual moles of particles after dissociation}}{\text{Moles of formula units dissolved}}\]
Relation with degree of dissociation:
i = 1 + α(n − 1)
\[\alpha=\frac{i-1}{n-1}\]
Theorems and Laws [12]
Statement: The solubility of a gas in a liquid is directly proportional to the pressure of the gas over the solution.
S = KH⋅P
Where S = solubility (mol L⁻¹), P = pressure (bar), KH = Henry's law constant (mol L⁻¹ bar⁻¹).
Gases like NH₃ and CO₂ do NOT obey Henry's law (they react with water).
Statement: The partial vapour pressure of any volatile component of a solution is equal to the vapour pressure ofthe pure component multiplied by its mole fraction in the solution.
For a binary solution of two volatile components:
\[\mathrm{P}_{1}=\mathrm{P}_{1}^{0}x_{1}\quad\mathrm{and}\quad\mathrm{P}_{2}=\mathrm{P}_{2}^{0}x_{2}\]
Raoult's law Using Dalton's law of partial pressures, Total vapour pressure (P) is given by:
\[\mathrm{P}=\mathrm{P}_{1}+\mathrm{P}_{2}=\mathrm{P}_{1}^{0}x_{1}+\mathrm{P}_{2}^{0}x_{2}=\left(\mathrm{P}_{2}^{0}-\mathrm{P}_{1}^{0}\right)x_{2}+\mathrm{P}_{1}^{0}\]
Composition of vapour phase: If y1 and y2 are the mole fractions of the components 1 and 2, respectively, in the vapour phase; then using Dalton's law of partial pressures: P1 = y1P and P2 = y2P
State Raoult’s law for solutions containing volatile solutes.
For a solution of volatile liquids, the partial vapour pressure of each component in the solution is directly proportional to its mole fraction.
If pi is the partial vapour pressure of ith component in a solution and χi is its mole fraction in the solution, then according to Raoult’s law,
pi ∝ χi
or pi = constant × χi ...(i)
If we consider the component in its pure state, then
`p_i = p_i^circ` and
χi = 1
Where Pi is the vapour pressure of the ith component in the pure state. In the case of the pure ith component, we shall have a 100% pure component, and χi will be equal to 1. Substituting the values in Eq. (i), we have
pi = constant × 1
or constant = `p_i^circ`
Again putting the value of the constant in Eq. (i), we have
`p_i = p_i^circ* chi_i`
State Raoult’s law.
Raoult’s law states that the partial vapour pressure of any volatile component of a solution is equal to the vapour pressure of the pure component multiplied by its mole fraction in the solution.
For a solution of volatile liquids, the partial vapour pressure of each component in the solution at a particular temperature is equal to the product of the vapour pressure of the component in the pure state and its mole fraction in the solution.
Mathematically, for a binary solution:
\[\ce{P_A = P{^{\circ}_{A}}\chi_A}\] and
\[\ce{P_B = P{^{\circ}_{B}}\chi_B}\]
The total vapour pressure is:
\[\ce{P_{total} = P_A + P_B = P{^{\circ}_{A}}\chi_A + P{^{\circ}_{B}}\chi_B}\]
State Raoult’s law for the solutions of non-volatile solutes in volatile solvents.
The vapour pressure of a solution containing a non-volatile solute at a particular temperature is directly proportional to the mole fraction of the solvent in the solution.
According to Raoult’s law,
`P = P_"solvent" = P^circ * chi_"solvent"` ...(i)
For a binary mixture,
`chi_"solute" + chi_"solvent" = 1`
∴ `chi_"solvent" = 1 - chi_"solute"`
Putting this value in the equation (i), we have
`P = P^circ xx (1 - chi_"solute")`
or `P/P^circ = 1 - chi_"solute"`
or `1 - P/P^circ = chi_"solute"`
or `(P^circ - P)/P^circ = chi_"solute"`
In the above equation, the term (P° − P) represents the lowering of vapour pressure in the formation of the solution. The term `(P^circ - P)/P^circ`is called the relative lowering of vapour pressure of the solution.
Statement:
The elevation in boiling point of a solvent is directly proportional to the molality of the solution.
Mathematical Expression:
ΔTb = Kbm
Where:
Kb = Ebullioscopic constant
Statement:
The depression in freezing point of a solvent is directly proportional to the molality of the solution.
Mathematical Expression:
ΔTf = Kf m
Where:
Kf = Cryoscopic constant
Statement:
The osmotic pressure of a dilute solution is directly proportional to its molar concentration at constant temperature.
Mathematical Expression:
π = MRT
Where:
π = Osmotic pressure
M = Molarity
R = Gas constant
T = Temperature in Kelvin
Statement:
At constant temperature, the solubility of a gas in a liquid is directly proportional to the pressure of the gas above the solution.
Mathematical Expression:
S ∝ P
S = KHP
Where:
S = Solubility of gas
P = Pressure of gas
KH = Henry’s law constant
Statement:
The total pressure exerted by a mixture of non-reacting gases is equal to the sum of the partial pressures of individual gases.
Mathematical Expression:
P = P1 + P2
Statement:
At constant temperature, the partial vapour pressure of a volatile component in a solution is equal to the product of its mole fraction and vapour pressure in pure state.
Mathematical Expression:
P1 = P10 x1
For binary solution:
P = P1 + P2
Statement:
For dilute solutions, the relative lowering of vapour pressure is equal to the mole fraction of the solute.
Mathematical Expression:
\[\frac{\Delta P}{P^0}=x_2\]
Key Points
Based on the physical states of solute and solvent, there are 9 types of solutions:
| S.No. | Solute | Solvent | Example |
|---|---|---|---|
| 1 | Solid | Liquid | Sea water, sugar in water, benzoic acid in benzene |
| 2 | Solid | Solid | Alloys — brass, bronze |
| 3 | Solid | Gas | Iodine in air |
| 4 | Liquid | Liquid | Ethanol in water, gasoline |
| 5 | Liquid | Solid | Amalgam (mercury in silver) |
| 6 | Liquid | Gas | Chloroform in nitrogen |
| 7 | Gas | Liquid | Carbonated water (CO₂ in water), O₂ in water |
| 8 | Gas | Solid | H₂ in palladium |
| 9 | Gas | Gas | Air (O₂, N₂, Ar mixture) |
Based on the amount of solute dissolved at a given temperature:
| Solution | Description |
|---|---|
| Unsaturated | Contains less solute than can be dissolved at given temperature |
| Saturated | Contains the maximum (equilibrium) quantity of solute at that temperature |
| Supersaturated | Contains more solute than the equilibrium amount (unstable) |
Factors Affecting Solubility
| Factor | Effect |
|---|---|
| Nature of solute & solvent | "Like dissolves like" — similar chemical character dissolves easily |
| Temperature (solid in liquid) | Endothermic: solubility increases with T; Exothermic: solubility decreases with T |
| Temperature (gas in liquid) | Solubility decreases with increase in temperature |
| Pressure (solid/liquid in liquid) | No effect (incompressible) |
| Pressure (gas in liquid) | Solubility increases with increasing pressure |
Electrolytes dissociate in solution → produce more particles → colligative properties are greater than expected. Observed molar masses are less than formula mass.
van't Hoff factor (i):
\[i=\frac{\text{Colligative property of electrolyte solution}}{\text{Colligative property of non-clectrolyte solution of same concentration}}\]
\[i=\frac{(\Delta T_f)_o}{(\Delta T_f)_t}=\frac{(\Delta T_b)_o}{(\Delta T_b)_t}=\frac{(\Delta P)_o}{(\Delta P)_t}=\frac{\pi_o}{\pi_t}=\frac{M_{\text{theoretical}}}{M_{\mathrm{observed}}}\]
| Type | Value of i | Example |
|---|---|---|
| Non-electrolyte | i = 1 | Glucose, urea |
| Electrolyte (dissociates) | i > 1 | NaCl (i ≈ 2), CaCl₂ (i ≈ 3) |
| Strong electrolyte | i = n (no. of ions after dissociation) | — |
| Weak electrolyte | Degree of dissociation α = (i − 1)/(n − 1) | — |
Modified Colligative Expressions:
\[\Delta P=i\cdot P_1^0\cdot x_2\]
\[\Delta T_{b}=iK_{b}m=\frac{i1000K_{b}w_{2}}{M_{2}W_{1}}\]
\[\Delta T_{f}=iK_{f}m=\frac{i1000\times K_{f}\times w_{2}}{M_{2}W_{1}}\]
\[\pi=i\cdot MRT=\frac{i\cdot W_{2}RT}{M_{2}V}\]
- Solutions of electrolytes exhibit colligative properties, but they do not obey the same quantitative relations as nonelectrolyte solutions.
- The observed colligative properties of electrolyte solutions are greater than those of nonelectrolyte solutions of the same concentration.
- The molar masses of electrolytes determined from colligative property measurements are found to be lower than their expected formula masses.
- The abnormal behaviour of electrolytes is due to their dissociation into two or more ions in aqueous solution, which increases the number of solute particles.
Important Questions [24]
- Define isotonic solutions.
- Define Boiling Point
- Derive Van’T Hoff General Solution Equation
- Henry's constant for CHA3BrA(g) is 0.159 mol dm−3 bar−1 25°C. Calculate its solubility in water at 25°C, if its partial pressure is 0.164 bar.
- Henry's law constant for CH3Br(g) is 0.159 mol dm–3 bar–1 at 25°C. What is the solubility of CH3Br(g) in water at the same temperature and partial pressure of 0.164 bar?
- A mixture of benzene and toluene contains 30% by mass of toluene. At 30°C, vapour pressure of pure toluene is 36.7 mm Hg and that of pure benzene is 118.2 mm Hg. Assuming that the two liquids form
- Calculate the mole fraction of solute, if the vapour pressure of pure benzene at a certain temperature is 640 mmHg and the vapour pressure of a solution of a solute in benzene is 600 mmHg.
- Define ebullioscopic constant.
- How will you determine the molar mass of non-volatile solute by elevation of boiling point?
- Derive the equation for the molar mass of solute from freezing point depression.
- Write the SI unit of the cryoscopic constant.
- Answer the following in one or two sentences. What is osmotic pressure?
- Define Osmosis.
- Derive an expression to calculate molar mass of non-volatile solute by osmotic pressure measurement.
- Define osmotic pressure.
- Define Semipermeable Membrane. OR Explain the term semipermeable membrane.
- Which of the Following is Not a Colligative Property?
- Define the Following Term: Isotonic Solution
- Define the following term: Hypotonic solution
- Choose the most correct option. In calculating osmotic pressure the concentration of solute is expressed in _______.
- Which of the following 0.1 M aqueous solutions will exert the highest osmotic pressure?
- How will you determine molar mass of solute from osmotic pressure?
- Write the condition of reverse osmosis.
- Derive the relationship between the degree of dissociation of an electrolyte and van’t Hoff factor.
Concepts [12]
- Introduction to Solutions
- Types of Solutions
- Capacity of Solution to Dissolve Solute
- Solubility
- Vapour Pressure of Solutions of Liquids in Liquids
- Colligative Properties of Nonelectrolyte Solutions
- Vapour Pressure Lowering
- Boiling Point Elevation
- Depression in Freezing Point
- Osmosis and Osmotic Pressure
- Colligative Properties of Electrolytes
- Overview of Solutions
