(English Medium)
Academic Year: 2025-2026
Date: मार्च 2026
Advertisements
- Answers to this paper must be written on the paper provided separately.
- You will not be allowed to write during first 15 minutes.
- This time is to be spent in reading the question paper.
- The time given at the head of this paper is the time allowed for writing the answers.
- Section A is compulsory. Attempt any four questions from Section B.
- The intended marks for questions or parts of questions are given in brackets [ ].
Which of the following will dissociate in aqueous solution to give a positive ion other than hydronium ion and a negative ion other than hydroxyl ion?
KOH
dil. HCl
NaCl
CH3COOH
Chapter:
A compound P is heated in a test tube with sodium hydroxide solution. A red litmus paper held at the mouth of the test tube turns blue.
Which of the following could compound P be?
Zinc sulphate
Copper sulphate
Ferrous sulphate
Ammonium sulphate
Chapter:
Assertion (A): Aqueous solution of potassium chloride can conduct electricity.
Reason (R): Conduction of electric current is due to the presence of free ions.
(A) is true and (R) is false.
(A) is false and (R) is true.
Both (A) and (R) are true and (R) is the correct explanation of (A).
Both (A) and (R) are true, but (R) is not the correct explanation of (A).
Chapter:
Identify the ion that contains one lone pair of electrons.
OH−1
H3O+
\[\ce{NH^+_4}\]
H+
Chapter:
Four reactions are shown below in the diagram:
 |
|
| 1. | 2. |
 |
 |
| 3. | 4. |
Which reactions produce water?
1 and 2
1 and 3
3 and 4
2 and 3
Chapter:
When compound X reacts with dilute sulphuric acid, it releases a gas that turns acidified potassium dichromate solution from orange to green. Which of the following could be compound X?
Lead nitrate
Copper carbonate
Sodium chloride
Potassium sulphite
Chapter:
The volume occupied by 2 moles of a gas at STP is ______.
22.4 L
2.24 L
44.8 L
4.48 L
Chapter:
Identify from the following metal oxides which can react with an acid as well as an alkali.
Silver oxide
Calcium oxide
Copper(II) oxide
Aluminium oxide
Chapter:
The structures of four hydrocarbons are shown below:
| \[\begin{array}{cc} \phantom{.....}\ce{CH3}\\ \phantom{...}|\\ \ce{H3C - C - H}\\ \phantom{...}|\\ \phantom{.....}\ce{CH3} \end{array}\] |
\[\begin{array}{cc} \ce{CH3}\phantom{.....}\\ |\phantom{.......}\\ \ce{C = CH2}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
\[\begin{array}{cc} \ce{H}\phantom{.....}\\ |\phantom{.....}\\ \ce{H3C - C - C = CH2}\\ |\phantom{....}|\\ \ce{H\phantom{...}H} \end{array}\] |
\[\begin{array}{cc} \phantom{...}\ce{CH3}\\ |\\ \ce{H3C - C = CH2} \end{array}\] |
How many isomers of butene are shown in the above structures?
1
2
3
4
Chapter:
Which element amongst the following has the largest atomic radius?
Al
S
Mg
Na
Chapter:
For which pH change is there the maximum increase in acidity?
| Initial pH | Final pH |
| 1 | 3 |
| Initial pH | Final pH |
| 2 | 6 |
| Initial pH | Final pH |
| 3 | 1 |
| Initial pH | Final pH |
| 6 | 2 |
Chapter:
The equation below shows the reaction between element ‘X’ and dilute sulphuric acid.
\[\ce{X_{(s)} + H2SO4_{(aq)} -> XSO4_{(aq)} + H2_{(g)}}\]
Which particles are responsible for conducting electricity in dilute sulfuric acid and compound XSO4?
Electrons
Only positive ions
Only negative ions
Both positive and negative ions
Chapter: [13] Practical Work
Methanol and ethanol belong to the same homologous series.
What does this statement mean?
Their molecules contain atoms only of carbon and hydrogen.
Their molecules have the same number of carbon atoms.
They have the same functional group.
They have the same relative molecular mass.
Chapter:
The ratio between the volumes occupied by 22 grams of carbon dioxide and 10 grams of hydrogen gas is ______.
2.2 : 1
1 : 2.2
1 : 10
10 : 1
Chapter:
In the process of electrorefining copper, as shown in the diagram below, which of the following statements is correct?

The anode is made of pure copper.
The cathode is made of impure copper.
Copper is deposited at the anode.
Copper ions from the anode move to the cathode and get deposited as pure copper.
Chapter:
Electroplating steel objects with silver involves a three-step process.
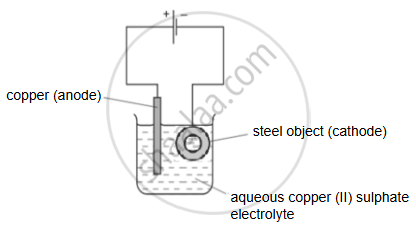
Step 1: A coating of copper is applied to the object.
Step 2: A coating of nickel is applied to the object.
Step 3: The coating of silver is applied to the object.
(a) A diagram of the apparatus used for step 1 is shown.
- The chemical process taking place on the surface of the object is \[\ce{Cu^2+_{ (aq)} + 2e- ->Cu_{(s)}}\].
What is the observation seen on the surface of the object? - Explain why the concentration of copper ions in the electrolyte remains constant throughout step 1.
(b) Give two changes which would be needed in order to coat nickel onto the object in step 2.
(c) Write down the reaction taking place at the positive electrode during step 3.
Chapter:
Identify the following:
A bond formed between two atoms by sharing of a pair of electrons, with both electrons being provided by the same atom.
Chapter: [2] Chemical Bonding
Identify the following:
A salt formed by the complete neutralization of an acid by a base.
Chapter: [3.2] Salts and their Preparations
Identify the following:
A reaction in which the hydrogen of an alkane is replaced by a halogen.
Chapter:
Identify the following:
The energy required to remove an electron from a neutral gaseous atom.
Chapter: [1] Periodic Table, Periodic Properties and Variations of Properties
Identify the following:
A homogenous mixture of two or more metals or a metal and a non-metal in a definite proportion in their molten state.
Chapter: [7.4] Alloys
When dilute sulphuric acid reacts with zinc granules, the gas evolved is ______, which can be tested using a burning splint.
hydrogen
carbon dioxide
Chapter:
A solution of copper(II) sulphate in sodium hydroxide solution forms a ______ precipitate.
pale blue
green
Chapter:
In methane, each hydrogen atom share(s) ______ electron(s) with the central carbon atom to complete its valence shell.
one
two
Chapter:
The electron affinity of element X is greater than that of element Y. The oxidising power of X is likely to be ______ than that of element Y.
more
less
Chapter:
The naturally occurring compound of a metal from which the metal can be extracted is called its ______.
ore
mineral
Chapter:
Match the following from Column A with Column B.
| Column A | Column B |
| (a) Aluminium | 1. Covalent compound |
| (b) Sulphuric acid | 2. Carbonate ore |
| (c) Calcination | 3. Hall Heroult’s process |
| (d) Calcium Chloride | 4. Contact Process |
| (e) Carbon tetrachloride | 5. Electrovalent compound |
Chapter:
Give the IUPAC name of the following organic compound:
\[\begin{array}{cc}
\ce{\phantom{..}H\phantom{..}H\phantom{..}OH}\\
|\phantom{...}|\phantom{...}|\\
\ce{H-C-C-C-H}\\
|\phantom{...}|\phantom{...}|\\
\ce{H\phantom{..}H\phantom{..}H}
\end{array}\]
Chapter:
Give the IUPAC name of the following organic compound:
\[\begin{array}{cc}
\ce{H\phantom{..}H\phantom{..}H\phantom{..}H\phantom{..}H}\\
|\phantom{...}|\phantom{...}|\phantom{...}|\phantom{...}|\\
\ce{H-C=C-C-C-C-H}\\
\phantom{.......}|\phantom{...}|\phantom{...}|\\
\ce{\phantom{.......}H\phantom{..}H\phantom{..}H}
\end{array}\]
Chapter:
Draw the structural formula for the following:
But-2-yne
Chapter:
Advertisements
Draw the structural diagram for the following compound:
1, 1, 1-trichloromethane
Chapter:
Draw the structural diagram for the following compound.
Pentan-2-ol
Chapter:
Give one significant observation when excess of chlorine gas reacts with ammonia.
Chapter:
State observation:
Zinc nitrate crystals are strongly heated.
Chapter:
Give reasons:
When ammonia gas is passed over black copper oxide in a combustion tube, a reddish-brown substance is left behind.
Chapter:
Give reasons:
Quick lime is not used to dry hydrogen chloride gas.
Chapter:
The electron affinity of an element X is greater than that of element Y.
How is the oxidising power of X likely to compare with that of Y?
Chapter: [1] Periodic Table, Periodic Properties and Variations of Properties
The electron affinity of an element X is greater than that of element Y.
How is the electronegativity of X likely to compare with that of Y?
Chapter: [1] Periodic Table, Periodic Properties and Variations of Properties
The electron affinity of an element X is greater than that of element Y.
State whether X is likely to be placed to the left or to the right of Y in the periodic table.
Chapter: [1] Periodic Table, Periodic Properties and Variations of Properties
State your observation of the following case:
When calcium hydroxide is heated with ammonium chloride crystals.
Chapter: [8] Study of Compounds A. Hydrogen Chloride
Write a balanced chemical equation for the following reaction:
Nitric acid reacts with zinc carbonate.
Chapter:
Write a balanced chemical equation for the following reaction:
Concentrated sulphuric acid is added to hydrated copper sulphate.
Chapter:
Name the main constituent metal in the following alloys:
Duralumin
Chapter: [7.4] Alloys
Name the metals present in the following alloys:
Brass
Chapter:
Write the balanced chemical equation for the laboratory preparation of hydrogen chloride gas.
Chapter:
Write a balanced chemical equation for the following:
Bromine gas is passed over ethene in the presence of carbon tetrachloride.
Chapter:
| Abhishek was given a salt ‘X’ which was white in colour for analysis. On strong heating, it produced a yellow residue, a colourless gas and also a reddish-brown gas. The solution of the salt ‘X’, when tested with an excess of ammonium hydroxide, produced a chalky white insoluble precipitate. |
- Name the coloured gas that evolved when Abhishek heated the salt strongly.
- Which cation was present in the sample given to Abhishek?
- Identify the salt given to Abhishek for analysis.
Chapter: [4] Analytical Chemistry
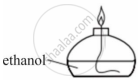
In a round bottom flask, a mixture of ethanol, acetic acid and concentrated sulphuric acid was heated.
- Name the type of reaction occurring in the above setup.
- What is the role of sulphuric acid in this reaction?
- State one observation that takes place during the reaction.
Chapter:
Identify the reactant and write the balanced equation for the following:
Nitric acid reacts with compound Q to give a salt Ca(NO3)2, water and carbon dioxide.
Chapter: [10] Study of Compounds C. Nitric Acid
What will be the mass of carbon dioxide that will contain the same number of molecules as present in 3.2 g of oxygen gas? [At. wt: O = 16, C = 12]
Chapter:
State the property exhibited by sulphuric acid in the following reaction:
Sulphur with concentrated sulphuric acid.
Chapter:
State the property exhibited by sulphuric acid in the following reaction:
Conversion of ferrous sulphide to hydrogen sulphide gas using sulphuric acid.
Chapter:
State the property exhibited by sulphuric acid in the following reaction:
Ethanol with concentrated sulphuric acid.
Chapter:
Write the equation of the following laboratory preparations:
Ethyne from calcium carbide.
Chapter:
Give a balanced equation for the following:
Conversion of acetic acid to ethyl acetate.
Chapter:
Give a balanced equation for the following:
Laboratory preparation of nitric acid.
Chapter:
Advertisements
A student was asked to perform an experiment in the laboratory based on the instructions given:
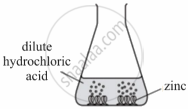
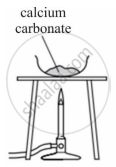
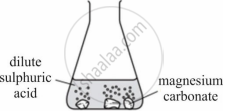
Observe the picture given below and state one observation for the experiment you would notice on mixing the given solution.
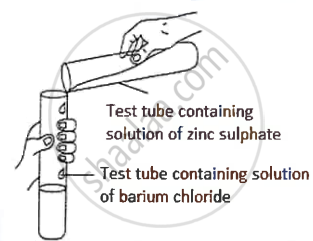
Chapter: [4] Analytical Chemistry
A student was asked to perform an experiment in the laboratory based on the instructions given:
Observe the picture given below and state one observation for the experiment you would notice on mixing the given solution.
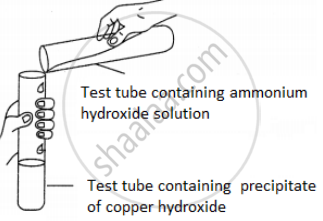
Chapter: [4] Analytical Chemistry
You are provided with the list of chemicals mentioned below in the box:
| Sodium hydroxide solution, copper carbonate, zinc, hydrochloric acid, copper, dilute sulphuric acid, chlorine, iron |
Using suitable chemicals from the list given, write a balanced chemical equation for the preparation of the salt mentioned below:
Copper sulphate
Chapter: [3.2] Salts and their Preparations
You are provided with the list of chemicals mentioned below in the box:
| Sodium hydroxide solution, copper carbonate, zinc, hydrochloric acid, copper, dilute sulphuric acid, chlorine, iron |
Using suitable chemicals from the list given, write a balanced chemical equation for the preparation of the salt mentioned below:
Sodium zincate
Chapter: [3.2] Salts and their Preparations
Solid ammonium dichromate decomposes as under:
\[\ce{(NH4)2Cr2O7 ->N2 + Cr2O3 + 4H2O}\]
If 126 g of ammonium dichromate decomposes, calculate:
- the number of moles of ammonium dichromate that undergoes decomposition.
- the mass of chromic oxide formed at the same time.
- the volume of nitrogen gas evolved at STP.
[At. wt: N = 14, Cr = 52, O = 16, H = 1]
Chapter:
Identify the reactant P in the following reaction:
\[\ce{Copper oxide + P → Copper + water}\]
Chapter:
Identify the reactant Q in the following reaction:
\[\ce{Iron pyrite + Q → Iron oxide + Sulphur dioxide}\]
Chapter:
Identify the reactant R in the following reaction:
\[\ce{Sodium chloride + R → Sodium nitrate + Silver chloride}\]
Chapter:
Give reasons for the following:
Nitric acid does not normally liberate hydrogen gas when it reacts with active metals.
Chapter:
Give reasons for the following:
Silver-plated cutlery is not considered as pure silver.
Chapter:
The following question relate to the extraction of aluminium by electrolysis.
Name the other aluminium containing compound added to alumina.
Chapter:
The following question relate to the extraction of aluminium by electrolysis.
Give the equation for the reaction that takes place at the cathode.
Chapter:
Write balanced chemical equation for action of warm water on AIN.
Chapter:
Give a balanced equation for the following:
Oxidation of carbon with conc. nitric acid.
Chapter: [10] Study of Compounds C. Nitric Acid
Give a balanced equation for the following:
Laboratory preparation of ethanol by using chloroethane and aqueous sodium hydroxide.
Chapter:
Rohit has solution X, Y and Z that has pH 2, 7 and 13 respectively.
Which solution will liberate sulphur dioxide gas when heated with sodium sulphite?
Chapter:
Rohit has solution X, Y and Z that has pH 2, 7 and 13 respectively.
Which solution will liberate ammonia gas when reacted with ammonium chloride?
Chapter:
Rohit has solution X, Y and Z that has pH 2, 7 and 13 respectively.
Which solution will not have any effect on litmus paper?
Chapter:
State giving reasons if zinc and aluminium can be distinguished by heating the metal powder with a concentrated sodium hydroxide solution.
Chapter:
State giving reasons if calcium nitrate and lead nitrate can be distinguished by adding ammonium hydroxide solution to the salt solution.
Chapter: [13] Practical Work
Draw the electron dot structure for the following:
Ammonium ion
[At. no.: N = 7, H = 1]
Chapter:
Write balanced chemical equations for the following conversions (A to C):
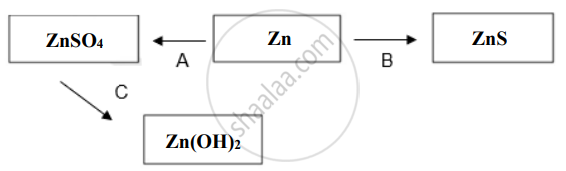
Chapter:
L, M and N are three elements with atomic numbers 13, 7 and 10 respectively. Answer the following question using only the alphabets given. Do not identify the elements.
Which element can combine with hydrogen to form a gas which produces dense white fumes with concentrated HCl?
Chapter:
L, M and N are three elements with atomic numbers 13, 7 and 10 respectively. Answer the following question using only the alphabets given. Do not identify the elements.
Which element has zero electron affinity?
Chapter:
L, M and N are three elements with atomic numbers 13, 7 and 10 respectively. Answer the following question using only the alphabets given. Do not identify the elements.
Which element can form an ionic compound with oxygen?
Chapter:
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CISCE previous year question papers ICSE Class 10 Chemistry with solutions 2025 - 2026
Previous year Question paper for CISCE ICSE Class 10 -2026 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Chemistry, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CISCE ICSE Class 10 .
How CISCE ICSE Class 10 Question Paper solutions Help Students ?
• Question paper solutions for Chemistry will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.