English Medium
Academic Year: 2025-2026
Date & Time: 25th February 2026, 10:30 am
Duration: 3h
Advertisements
General Instructions:
Read the following instructions carefully and follow them:
- This question paper contain 39 questions. All questions are compulsory.
- The question paper is divided into three sections - A, B and C.
Section A: Biology (30 marks)
Section B: Chemistry (25 marks)
Section C: Physics (25 marks) - The question paper has MCQs, VSAs, SAs, LAs and CBQs. Marks are given against each question.
- There are case based questions (CBQs) with three sub-questions and are of 4 marks each.
- Divide your answer sheet into three sections as per the question paper - Section A (Biology), Section B (Chemistry) and Section C (Physics). It is compulsory to answer each question in its respective section. Do not mix answers of one section into the other section.
- Instructions are given with each section and question, wherever necessary.
- Kindly note that a separate question paper has been provided for visually impaired candidates.
- There is no overall choice in the question paper. However, an internal choice has been provided in a few questions. Only one of the choices in such questions
must be attempted.
Identify the correct statement for spirogyra, leishmania and hydra:
they reproduce sexually.
they are unicellular.
they are multicellular.
they reproduce asexually.
Chapter:
Which structure in a leaf is mainly responsible for gaseous exchange?
Xylem
Stomata
Phloem
Cuticle
Chapter:
Pancreas secretes pancreatic juice which contain certain enzyme that helps in digestion of food.
Choose the correct option from the following:
Trypsin digests emulsified fats and lipase digests proteins.
Trypsin digests proteins and lipase digests emulsified fats.
Trypsin and lipase both digests fats.
Trypsin digests proteins and lipase digests carbohydrates.
Chapter:
From the given situations, identify ‘Chemotropic’ and ‘Geotropic’ movements in parts of plants, respectively:
- Growth of pollen tube towards ovule.
- Movement of sunflower towards sunlight.
- Movement of root towards Earth/Gravity.
- Movement of leaves due to breeze.
Choose the correct option:
(i) and (iii) respectively
(iii) and (i) respectively
(i), (ii) and (iii), (iv) respectively
(i), (iii) and (ii), (iv) respectively
Chapter:
Which of the following group is not ‘biodegradable’?
Vegetable peels, dead leaves, paper
Cow dung, leather bag, water
Polythene bag, rubber band, ball pen
Paper, fruits, bones
Chapter:
Study the given diagram of the heart, with blood vessels marked ‘1’ and ‘2’.
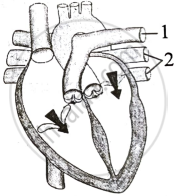
From the following statements, establish the relationship between heart and/or the two blood vessels:
- Blood vessel 1 - It carries carbon dioxide rich blood to the lungs.
- Blood vessel 2 - It carries oxygen rich blood from the lungs.
- Blood vessel 2 - Left atrium relaxes as it receives blood from this blood vessel.
- Blood vessel 1 - Right atrium has thick wall as it has to pump blood to this vessel.
The option with correct statements is:
(i) and (ii)
(ii) and (iii)
(ii), (iii) and (iv)
(i), (ii) and (iii)
Chapter:
Plants use variety of techniques to get rid of their waste materials. Some are mentioned below. Identify the incorrect one:
Excess water is given out by transpiration.
Gums and Resins are wastes that are stored.
Roots secrete some wastes into the soil.
Flowers can store some waste products.
Chapter:
Assertion (A): Reflex actions do not involve thinking.
Reason (R): Most reflex actions are controlled by the spinal cord.
Both, Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
Both, Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (А).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Assertion (A): Ozone at higher levels of atmosphere is a product of UV radiation acting on oxygen molecule.
Reason (R): The higher energy of UV splits apart some molecular O2.
Both, Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
Both, Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Bile juice contains no digestive enzymes, yet it is important for digestion. Why?
Chapter:
Name some substances present in initial filtrate and subsequently selectively reabsorbed in the tubules of nephron.
Chapter:
Draw a neat diagram to show germination of pollen on the female reproductive part of the flower. Name and label only the following parts:
- The part that receives the pollen grain.
- The structure that carries the male germ cell to reach the female germ cell.
Chapter:
State two differences between the act of chewing food and salivation on sight of food.
Chapter:
List any two differences between pollination and fertilization.
Chapter: [7] How do Organisms Reproduce?
State the difference between nephron and neuron.
Chapter:
Differentiate between the following pair of terms:
Motor nerve and sensory nerve.
Chapter:
Give difference between the following:
Consumers and decomposers
Chapter:
Given below is a pyramid showing various trophic levels in an ecosystem:
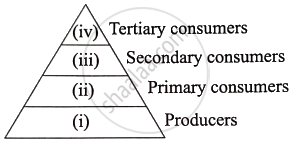
- From the organisms listed below, identify which one is to be placed at which trophic level?
Deer, Grass, Lion, Snake, Rabbit - Discuss the reason why primary consumers will have more energy as compared to secondary consumers?
- Why is the base of the pyramid broad?
Chapter:
| Mendel took garden pea plants with different characteristics, such as height to study the inheritance pattern of factors (genes). He crossed tall pea plant with short pea plant and obtained all the tall plants in the F1 generation. |
Answer the following questions:
(a) Why only tall pea plants were observed in F1 progeny? [1]
(b) By which method did Mendel obtain F2 progeny? [1]
(c) (i) Write one difference between dominant and recessive trait. [2]
OR
(c) (ii) Write two observations made by Mendel about F1 progeny. [2]
Chapter:
Given below is a certain situation. Analyse it and describe its possible impact:
A population of bacteria living in temperate waters whose temperature increased by global warming.
Chapter:
Given below is a certain situation. Analyse it and describe its possible impact:
The sperm encounters the egg when it reaches the oviduct in human females.
Chapter:
Given below is a certain situation. Analyse it and describe its possible impact:
Self pollination does not occur in a flower that contains only pistil.
Chapter:
Advertisements
Given below is a certain situation. Analyse it and describe its possible impact:
Egg does not get fertilised in a human female.
Chapter:
Given below is a certain situation. Analyse it and describe its possible impact:
When the seed is placed under appropriate condition of water and air in the soil?
Chapter:
Given below is a certain situation. Analyse and describe what would happen when:
Spores are liberated from blob-like structures of the bread mould?
Chapter:
What happens when Bryophyllum leaf falls on the wet soil?
Chapter:
Given below is a certain situation. Analyse and describe what would happen when:
A pollen from different species land on the stigma of totally unrelated species?
Chapter:
Given below is a certain situation. Analyse and describe what would happen when:
Copper-T is placed in the uterus of a human female?
Chapter:
Given below are certain situations. Analyse and describe what would happen when:
Spirogyra breaks into smaller fragments upon maturation?
Chapter:
The gases evolved on heating lead (II) nitrate crystals are ______.
NO and O2
N2 and NO2
NO2 and H2
NO2 and O2
Chapter:
- \[\ce{AgNO3 + NaCl -> NaNO3 + AgCl}\]
- \[\ce{K2SO4 + BaCl2 -> BaSO4 + 2KCl}\]
Which of the following options clearly describes both the reactions?
(i) is double displacement, (ii) is displacement reaction.
Both, (i) and (ii) are displacement reactions and precipitation reactions.
Both, (i) and (ii) are double displacement reactions and precipitation reactions.
(i) is displacement, (ii) is double displacement reaction.
Chapter:
Which one of the following can be used as an acid-base indicator by a visually impaired (blind) student?
Turmeric
Vanilla essence
Methyl orange
Litmus
Chapter:
The hydrocarbons with general formula CnH2n represents ______.
alkane
alkene
alkyne
cyclic compounds
Chapter:
When an element ‘X’ reacts with water, it starts floating. Identify the element ‘X’:
Potassium
Calcium
Sodium
Iron
Chapter:
Which of the following is a poor conductor of electricity?
Pb
Cu
Ag
Al
Chapter:
The natural sources of oxalic acid, lactic acid and methanoic acid respectively are ______.
tomato, curd, ant-sting
tomato, orange, nettle-sting
orange, milk, ant-sting
orange, sour milk, nettle-sting
Chapter:
Assertion (A): Carbon shares its valence electrons with other atoms of carbon or with atoms of other elements.
Reason (R): The shared electrons belong to the outermost shells of both the atoms and lead to both atoms attaining the noble gas configuration.
Both, Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
Both, Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (А).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Why does one feels pain and irritation when stung by honey-bee? Rubbing of baking soda on the stung area gives relief. How?
Chapter:
What do you observe when iron nail is kept in copper sulphate solution for sometime.
Chapter:
What happens when potassium iodide solution is mixed with lead nitrate solution? Write balanced chemical equations to support your answer.
Chapter:
What is observed when silver chloride is exposed to sunlight? Give the type of reaction involved.
Chapter: [1] Chemical Reactions and Equations
Explain chlor-alkali process with chemical equation. Name the products formed at anode and cathode.
Chapter:
Write the preparation of the following compound with balanced chemical equation:
Baking Soda
Chapter:
How is bleaching powder prepared? Write a chemical equation of the reaction involved in the preparation of bleaching powder.
Chapter:
Write the preparation of the following compound with balanced chemical equation:
Plaster of Paris
Chapter:
Advertisements
Read the following passage and answer the questions given below:
| Most of metals occur in combined state in form of ores. Carbonate ores are converted into oxides by calcination and sulphide ores by roasting. Oxides are reduced with suitable reducing agent like carbon to get free metal. Highly reactive metals like - Al, Mg are also used as reducing agents to obtain metal from their oxides. Most reactive metals are obtained by electrolytic reduction of their molten ores. Alloying is a very good method of improving the properties of a metal. We can get desired properties by this method. The electrical conductivity and melting point of an alloy is less than that of pure metals. |
(a) Why carbonate or sulphide ores are converted to oxides before extraction of metal from it? [1]
(b) Write a reaction in which Aluminium is used as a reducing agent to obtain metal from its oxide. [1]
(c) (i) How is copper obtained from its ore (Cu2S)? Give equations of the reactions. [2]
OR
(c) (ii) (I) Why highly reactive metals cannot be obtained from their oxides by using carbon as a reducing agent? [1]
(II) Why solder, an alloy of lead and tin, is used for welding electrical wires together? [1]
Chapter:
Give reasons for the following:
Covalent compounds are poor conductor of electricity.
Chapter:
Give reasons for the following:
Soap does not form lather in hard water.
Chapter:
Give reasons for the following:
Carbon shows catenation but silicon does not.
Chapter:
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Chapter: [4] Carbon and its Compounds
Write the chemical equation for the following:
Hydrogenation of ethene.
Chapter:
Mohan heated ethanol with a compound ‘X’ in the presence of a few drops of conc. H2SO4 and observed a sweet smelling compound ‘Y’ is formed. When ‘Y’ is treated with sodium hydroxide it gives back ethanol and a compound ‘Z’.
- Identify ‘X’, ‘Y’ and ‘X’.
- Write the role of conc. H2SO4 in the reaction.
- Write the chemical equations involved and name the reactions.
Chapter:
A convex lens of focal length 15 cm, is forming a real image. If the size of image is same as the size of object, then position of object and position of inage will be, respectively ______.
−15 cm and −15 cm from lens
–15 cm and +15 cm from lens
–30 cm and +30 cm from lens
–30 cm and –30 cm from lens
Chapter:
When you look at an object very close to your eyes, the ______.
Ciliary muscles of your eye contract and the eye lens becomes thick.
Ciliary muscles of your eye get relaxed and the eye lens becomes thick.
Ciliary muscles of your eye contract and the eye lens becomes thin.
Ciliary muscles of your eye get relaxed and the eye lens becomes thin.
Chapter:
Assertion (A): When rays of white light pass through a prism, on emerging they give spectrum of seven colours.
Reason (R): It is due to the scattering of light that red light bends minimum and violet light bends the maximum.
Both, Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
Both, Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Draw the ray diagram for the image formation by a lens which shows a magnification of +2.
Chapter:
The resistance of a wire of 0.01 cm radius and 1.0 cm length is 7 Ω. Calculate its resistivity.
Chapter:
An electric heater is rated 220 V; 11 A. Calculate the power consumed if the heater is operated at 200 V.
Chapter:
The pattern of magnetic field due to a current carrying wire depends upon the shape made by that wire. Justify.
Chapter:
A current carrying straight wire AB is shown in the given diagram. Out of X, Y and Z on which point will the strength of magnetic field be maximum and why?
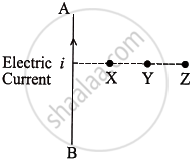
Chapter:
Describe an activity to show that a current carrying conductor, placed in an external magnetic field experiences a force.
Chapter:
Imagine that you are sitting in a chamber with your back to one wall. An electron beam, moving horizontally from back wall towards the front wall, is deflected by a strong magnetic field to your right side. What is the direction of magnetic field?
Chapter: [12] Magnetic Effects of Electric Current
With the help of a suitable ray diagram, explain how hypermetropia is corrected.
Chapter:
Read the following passage and answer the questions given:
| Lenses can form different types of images depending upon their focal length and position of object. A convex lens can create real, inverted or virtual, erect images, while a concave lens forms only virtual and diminished images. The focal length determines the power of lens. Convex lenses have positive focal length while concave lenses have negative focal length by convention. When lenses are placed together, their combined power is determined by the sum of their individual powers. Ray diagrams help to visualize how light converges or diverges through lens to form an image. |
(a) A convex lens of focal length 20 cm is used to form an image. If an object is placed at 40 cm from the lens, what will be the position and nature of image? [1]
(b) Illustrate the formation of image with the help of ray diagram, when the object is placed between the optical centre and principal focus of concave lens. [1]
(c) (i) A lens combination consists of a convex lens of focal length 30 cm and a concave lens of focal length 15 cm placed together. Find the equivalent focal length and power of this lens combination. [2]
OR
(c) (ii) Two lenses are placed in contact. One is a concave lens with focal length 2 m and the other is a convex lens with focal length 1.5 m. What type of lens will the combination behave as (convex or concave)? Give reason. [2]
Chapter:
Study the given electric circuit in which 2 A electric current is flowing between points X and Y.
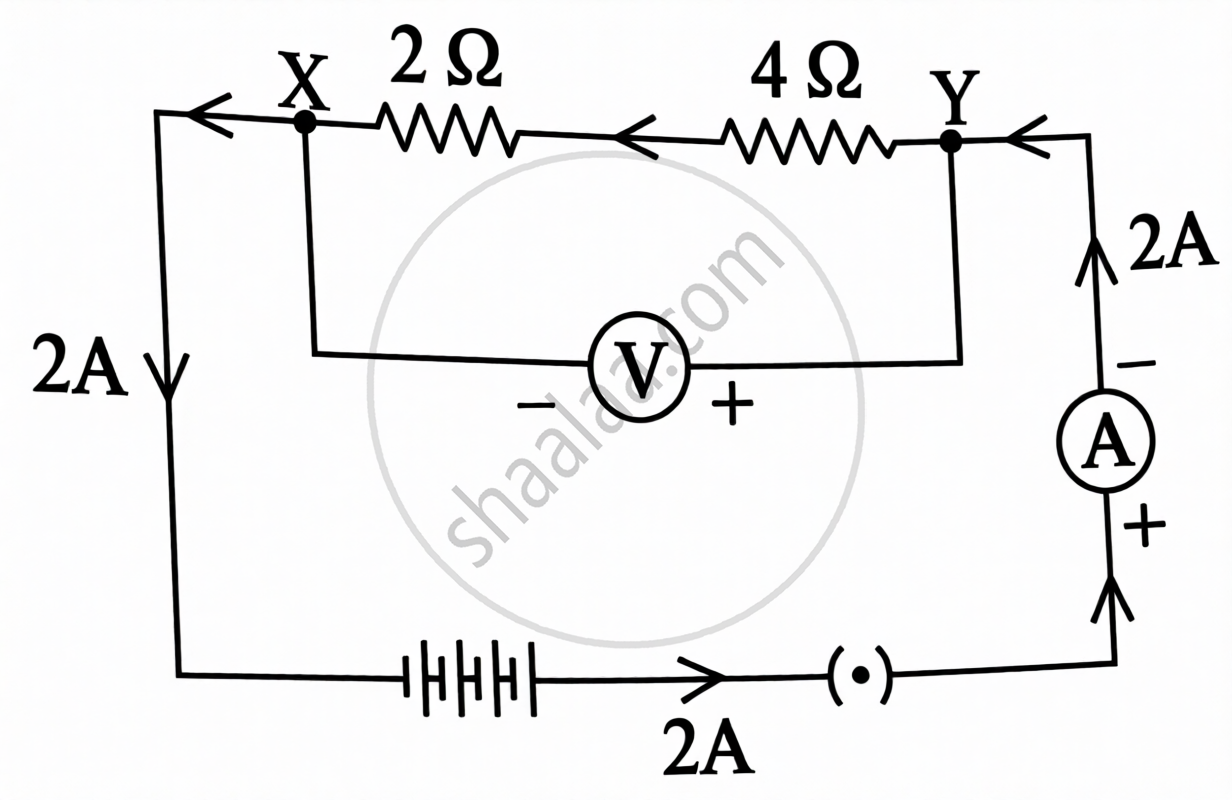
- Using the battery, key, voltmeter and ammeter in this given electric circuit, redraw a circuit diagram in which 2 Ω and 4 Ω resistors are connected between X and Y in parallel combination.
- Circuit drawn by you, in which resistors are connected in parallel combination, calculate the electric current flowing through 4 Ω resistor.
Chapter:
Study the given electric circuit in which 2 A electric current is flowing between points X and Y.

- Using the battery, key, voltmeter and ammeter in this given electric circuit, redraw a circuit diagram in which 2 Ω and 4 Ω resistors are connected between X and Y in parallel combination.
- Circuit drawn by you, in which resistors are connected in parallel combination, calculate the electric current flowing through 4 Ω resistor.
Chapter:
Two lamps ‘A’ and ‘B’ of rating 50 W; 220 V and 100 W, 220 V are connected in series combination. Find out the ratio of the resistances (RA : RB) of these lamps.
Chapter:
Derive the expression for the equivalent resistance of three resistors R1, R2 and R3 connected in parallel.
Chapter:
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CBSE previous year question papers Class 10 Science with solutions 2025 - 2026
Previous year Question paper for CBSE Class 10 Science-2026 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Science, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CBSE Class 10.
How CBSE Class 10 Question Paper solutions Help Students ?
• Question paper solutions for Science will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
