Advertisements
Advertisements
प्रश्न
Write the molecular formula of the first six members of homologous series of nitro – alkanes.
Advertisements
उत्तर
- \[\ce{CH2NO2}\] Nitromethane
- \[\ce{CH2-CH2NO2}\] Nitroethane
- \[\ce{CH3-CH2-CH2NO2 1-}\] nitropropane
- \[\ce{CH3-CH2-CH2-CH2-NO2 1-}\] nitrobutane
- \[\ce{CH3-CH2-CH2-CH2-CH2-NO2 1 –}\] nitropentane
- \[\ce{CH2-CH2-CH2-CH2-CH2-CH2-CH2-NO2 1-}\] nitrohexane
APPEARS IN
संबंधित प्रश्न
Find out all the functional groups present in the following polyfunctional compound.
Penicillin G, a naturally occurring antibiotic.
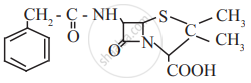
Identify primary, secondary, tertiary and quaternary carbon in the following compound.
Write a note on homologous series.
The reagent used to confirm the presence of five hydroxyl groups in glucose is ____________.
Which of the following yields only one type of monosubstituted chloroalkane upon chlorination?
How many 3° carbon atoms are present in 1, 4 dibromo-2-methylpentane?
An organic compound 'X' (molecular formula C6H7O2N) has six carbons in a ring system, two double bonds and also a nitro group as a substituent, 'X' is ______.
A pyranose ring consists of a skeleton of ______.
Identify primary, secondary, tertiary and quaternary carbon in the following compounds.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
\phantom{}|\phantom{....}|\phantom{...............}\\
\phantom{}\ce{CH3}\phantom{.}\ce{CH3}\phantom{.............}
\end{array}\]
Identify primary, secondary, tertiary, and quaternary carbon in the following compounds.
\[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
\phantom{}|\phantom{....}|\phantom{................}\\
\phantom{.}\ce{CH3\phantom{.}CH3}\phantom{..............}
\end{array}\]
