Advertisements
Advertisements
प्रश्न
Write equation for the reaction that will take place when copper sulphate solution is added to sodium hydroxide solution.
Advertisements
उत्तर
For the reaction that will take place when copper sulphate solution is added to sodium hydroxide solution the equation is as:
\[\ce{CuSO4 + 2NaOH -> Cu(OH)2 + Na2SO4}\]
APPEARS IN
संबंधित प्रश्न
Identify the cations in the following case:
NaOH solution, when added to the Solution (A), gives a reddish brown precipitate
Name a white, insoluble oxide that dissolves when fused with caustic soda or caustic potash.
Using sodium hydroxide solution, how will you distinguish: Zinc nitrate solution from calcium nitrate solution
Identify the substance Q based on the information given below:
The white crystalline solid Q is soluble in water. It liberates a pungent smelling gas when heated with sodium hydroxide solution.
Using Sodium hydroxide solution, how would you distinguish: Zinc nitrate solution from calcium nitrate solution.
Distinguish by adding:
Sodium hydroxide solution and ammonium hydroxide solution to calcium salt solution and lead salt solution.
Distinguish by adding:
Sodium hydroxide solution and ammonium hydroxide solution to Fe(II) salt solution and Fe(III) salt solution.
Identify the cation in the following case:
Sodium hydroxide solution when added to Solution C gives a white precipitate which is insoluble in excess of sodium hydroxide solution.
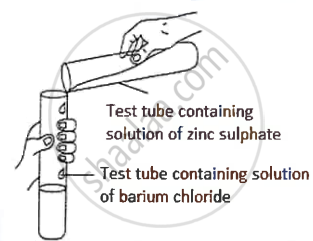
A student was asked to perform an experiment in the laboratory based on the instructions given:
Observe the picture given below and state one observation for the experiment you would notice on mixing the given solution.
Write the word equation when ammonium salt is heated with caustic soda solution.
