Advertisements
Advertisements
प्रश्न
Write balanced equation for the reaction of dilute sulphuric acid with the following:
Copper carbonate
Advertisements
उत्तर
Balanced equation is : Copper Carbonate
CuCO3 + H2SO4 → CuSO4 + H2O + CO2
APPEARS IN
संबंधित प्रश्न
State your observations when Concentrated Sulphuric acid is added to Sugar Crystals
State the conditions required for the given reaction to take place:
Any two conditions for the conversion of sulphur dioxide to sulphur trioxide
Name the following:
Products obtained by treating ferrous sulphide with dilute sulphuric acid.
Give reason for the following:
When barium chloride is added to dilute sulphuric acid, a white precipitate is formed.
What do you see when concentrated sulphuric acid is added to copper sulphate-5-water ( You are not required to say what is happing, nor is it necessary to name the products.)
Which concentrated acid oxidizes sulphur directly to sulphuric acid ? Write the equation for the same. What is the name of the process by which sulphuric acid is manufactured ? Name the catalyst used in the process.
Complete the following sentence, choosing the correct word from the given options.
"Concentrated sulphuric acid is used in the laboratory preparation of nitric acid and hydrochloric acid because it is ____________ in comparison to these two acids."
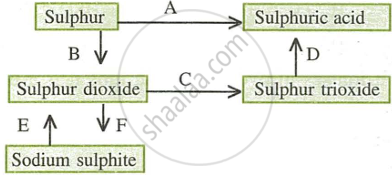
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
In the given equation identify the role played by concentrated sulphuric acid S + 2H2SO4→ 3SO2 + 2H2O
Write a balanced equation for the following conversion:
Lead sulphate from lead nitrate and sulphuric acid.
