Advertisements
Advertisements
प्रश्न
Which of the following graphs correctly represents the variation of a particle momentum with its associated de-Broglie wavelength?
पर्याय
Advertisements
उत्तर
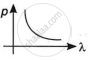
Explanation:
de-Broglie wavelength is the wavelength that is connected to an object in relation to its momentum and mass. Typically, the force of a particle is inversely proportional to its de-Broglie wavelength.
A photon's momentum is determined by:
P = `E/c = h/lambda` where E is the energy of the photon, c is the speed of light in vacuum, h is Planck's constant and λ is the de-Broglie wavelength.
According to de-Broglie, `p = h/lambda` or `p ∝ 1/lambda`.
By this relation, we can conclude that the linear momentum of a photon is inversely proportional to the de-Broglie wavelength. The graph of p vs λ shall be a rectangular hyperbola.
संबंधित प्रश्न
A proton and an α-particle have the same de-Broglie wavelength Determine the ratio of their speeds.
What is the de Broglie wavelength of a dust particle of mass 1.0 × 10−9 kg drifting with a speed of 2.2 m/s?
A particle is dropped from a height H. The de Broglie wavelength of the particle as a function of height is proportional to ______.
An electron is moving with an initial velocity `v = v_0hati` and is in a magnetic field `B = B_0hatj`. Then it’s de Broglie wavelength ______.
A proton and an α-particle are accelerated, using the same potential difference. How are the de-Broglie wavelengths λp and λa related to each other?
Assuming an electron is confined to a 1 nm wide region, find the uncertainty in momentum using Heisenberg Uncertainty principle (∆x∆p ≃ h). You can assume the uncertainty in position ∆x as 1 nm. Assuming p ≃ ∆p, find the energy of the electron in electron volts.
Given below are two statements:
Statement - I: Two photons having equal linear momenta have equal wavelengths.
Statement - II: If the wavelength of photon is decreased, then the momentum and energy of a photon will also decrease.
In the light of the above statements, choose the correct answer from the options given below.
How will the de-Broglie wavelength associated with an electron be affected when the velocity of the electron decreases? Justify your answer.
How will the de-Broglie wavelength associated with an electron be affected when the accelerating potential is increased? Justify your answer.
E, c and `v` represent the energy, velocity and frequency of a photon. Which of the following represents its wavelength?