Advertisements
Advertisements
प्रश्न
Which of the following graphs correctly represents the variation of a particle momentum with its associated de-Broglie wavelength?
विकल्प
Advertisements
उत्तर
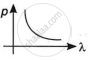
Explanation:
de-Broglie wavelength is the wavelength that is connected to an object in relation to its momentum and mass. Typically, the force of a particle is inversely proportional to its de-Broglie wavelength.
A photon's momentum is determined by:
P = `E/c = h/lambda` where E is the energy of the photon, c is the speed of light in vacuum, h is Planck's constant and λ is the de-Broglie wavelength.
According to de-Broglie, `p = h/lambda` or `p ∝ 1/lambda`.
By this relation, we can conclude that the linear momentum of a photon is inversely proportional to the de-Broglie wavelength. The graph of p vs λ shall be a rectangular hyperbola.
संबंधित प्रश्न
An electron and a photon each have a wavelength of 1.00 nm. Find
(a) their momenta,
(b) the energy of the photon, and
(c) the kinetic energy of electron.
For what kinetic energy of a neutron will the associated de Broglie wavelength be 1.40 × 10−10 m?
Obtain the de Broglie wavelength associated with thermal neutrons at room temperature (27°C). Hence explain why a fast neutron beam needs to be thermalised with the environment before it can be used for neutron diffraction experiments.
The energy and momentum of an electron are related to the frequency and wavelength of the associated matter wave by the relations:
E = hv, p = `"h"/lambda`
But while the value of λ is physically significant, the value of v (and therefore, the value of the phase speed vλ) has no physical significance. Why?
Describe briefly how the Davisson-Germer experiment demonstrated the wave nature of electrons.
When a light wave travels from air to glass ______.
Sodium and copper have work function 2.3 eV and 4.5 eV respectively. Then, the ratio of the wavelengths is nearest to ______.
A particle is dropped from a height H. The de Broglie wavelength of the particle as a function of height is proportional to ______.
Two particles A and B of de Broglie wavelengths λ1 and λ2 combine to form a particle C. The process conserves momentum. Find the de Broglie wavelength of the particle C. (The motion is one dimensional).
The ratio of wavelengths of proton and deuteron accelerated by potential Vp and Vd is 1 : `sqrt2`. Then, the ratio of Vp to Vd will be ______.