Advertisements
Advertisements
प्रश्न
What are the types of emission?
Advertisements
उत्तर
There are four types of emissions:
(i) Thermionic emission. (ii) Photoelectric emission.
(iii) Field emission. (iv) Secondary emission.
संबंधित प्रश्न
What is thermionic emission?
In the following fig. shows a radioactive source S in a thick lead container. The radiations pass through an electric field between the plates P and Q. Complete the diagram to show the paths of α , β and γ radiations.
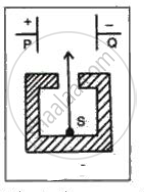
Why is the source S placed in a thick lead container?
A certain nucleus A (mass number 238 and atomic number 92) is radioactive and becomes a nucleus B (mass number 234 and atomic number 90) by the loss of one particle.
What particle was emitted?
State any two uses of cathode rays.
Why are materials of low work function preferred as thermionic cathode materials?
In a cathode ray tube state the purpose of covering cathode by thorium and carbon.
Justify with reason, whether the following nuclear reactions are allowed or not.
\[\ce{^A_Z X -> ^A_{Z + 1}Y + ^0_{-1}β}\]
In U238 ore containing Uranium the ratio of U234 to Pb206 nuclei is 3. Assuming that all the lead present in the ore is final stable product of U238. Half life of U238 to be 4.5 × 109 years and the age of ore is ______ × 109 years. (in 109 years)
