Advertisements
Advertisements
प्रश्न
The figure shows a process on a gas in which pressure and volume both change. The molar heat capacity for this process is C.

पर्याय
C = 0
C = Cv
C > Cv
C < Cv
Advertisements
उत्तर
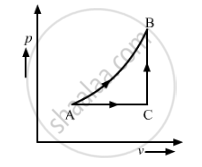
C > Cv
Consider two processes AB and ACB; let W be the work done. C is the molar heat capacity of process AB. Process ACB can be considered as the sum of the two processes, AC and CB. The molar heat capacity of process AC is Cp, as pressure is constant in this process and the molar heat capacity of process CB is Cv, as volume is constant in it.
Internal energy, U, is a state function, i.e. it doesn't depend on the path followed. Therefore,
UAB = UACB
WAB > WACB
Work done in the p-V diagram is the area enclosed under the curve.
⇒ WAB + UAB > WACB + UACB
⇒ C > CV + CP
Molar heat capacity is the heat supplied per mole to change the temperature by a degree Kelvin and according to the first law of thermodynamics, dQ = dU + dW, where dQ is the heat supplied to the system in a process.
⇒ C > CV
