Advertisements
Advertisements
प्रश्न
The diagram shows an experimental set up for the laboratory preparation of a pungent smelling gas. The gas is alkaline in nature.
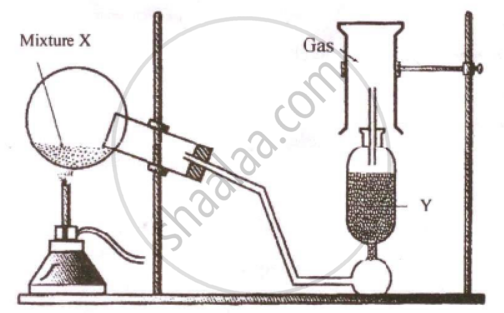
- Nature the gas collected in the jar.
- Write the balance equation for the above preparation.
- How is the gas being collected?
- Name the drying agent used.
- How will you find that the jar is full of gas?
The diagram below shows the set up for the laboratory preparation of a pungent alkaline gas.
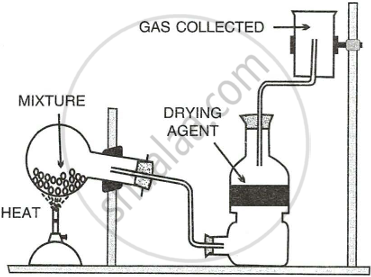
- Name the gas collected in the jar.
- Give a balanced equation for the above preparation.
- State how the above gas is collected.
- Name the drying agent used.
- State how you will find out that the jar is full of the pungent gas.
Advertisements
उत्तर
- Ammonia
- \[\ce{NH4Cl + Ca(OH)2 ->[Heat] CaCl2 + 2H2O + 2NH3↑}\]
- By downward displacement of air.
- Quicklime (CaO)
- Bring moist red litmus paper to the mouth of the inverted jar; it immediately turns blue.
Or
Bring a glass rod dipped in hydrochloric acid to the mouth of the inverted jar. If it produces dense white fumes, then the jar is full of gas.
संबंधित प्रश्न
Is ammonia more dense or less dense than air?
What property of ammonia is demonstrated by the fountain experiment?
List the properties of ammonia that make it
(i) A good refrigerant
(ii) A cleaning agent
(iii) As a source of hydrogen
Choose the correct word or phrase from the bracket to complete the following sentence:
Heating ammonium chloride with sodium hydroxide produces ______.
Choose the correct word or phrase from the bracket to complete the following sentences.
Heating solution of ammonium chloride with sodium nitrite produces ______
Name the other ion formed when ammonia dissolves in water
Name the gas evolved when the following mixtures are heated:
Sodium nitrate and ammonium chloride.
What do you observe when Ammonia is passed over heated lead oxide.
What do you observe when Ammonia comes in contact with the eyes of a person.
Differentiate between:
Aqueous ferrous and ferric sulphate solution
