Advertisements
Advertisements
प्रश्न
Differentiate between:
Aqueous ferrous and ferric sulphate solution
Advertisements
उत्तर
| Aqueous ferrous | Ferric sulphate solution |
|
When ammonium solution is added to aqueous solution of ferrous sulphate a dirty green precipi tate of ferrous hydroxide is obtained which is insoluble in excess of ammonium solution. Hydroxide. \[\ce{FeSO4 + 2NH4OH -> (NH4)2SO4 + Fe(OH)2}\] |
On adding ammonium solution to ferric sulphate solution we get ammonium sulphate and reddish brown precipi tate of ferric hydroxide which is insoluble in excess of ammonium solution . \[\ce{Fe2(SO4)3 + 6NH4OH -> 3(NH4)2SO4 + 2Fe(OH)3}\] |
APPEARS IN
संबंधित प्रश्न
Give two reactions to show reducing property of ammonia.
What is liquor ammonia fortis?
Choose the correct word or phrase from the bracket to complete the following sentences.
Heating solution of ammonium chloride with sodium nitrite produces ______
Given one test can be used to detect the presence of the ion produced
The diagram shows an experimental set up for the laboratory preparation of a pungent smelling gas. The gas is alkaline in nature.
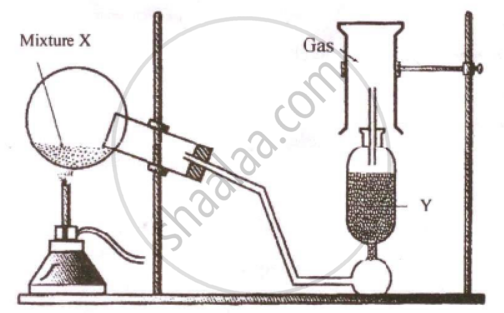
- Nature the gas collected in the jar.
- Write the balance equation for the above preparation.
- How is the gas being collected?
- Name the drying agent used.
- How will you find that the jar is full of gas?
What do you observe when Excess ammonia is mixed with chlorine.
What do you observe when Ammonium solution is added to ferric chloride solution.
What do you observe when Ammonia comes in contact with the eyes of a person.
