Advertisements
Advertisements
प्रश्न
State your observation When moist starch iodide paper is introduced into chlorine gas.
Advertisements
उत्तर
When moist starch iodide paper is introduced into chlorine gas, chlorine oxidizes iodide to iodine, which shows up as blue when complexed with starch.
APPEARS IN
संबंधित प्रश्न
State your observations when Dilute Hydrochloric acid is added to Copper carbonate
Select from the list the gas that matches the description given in case
[ammonia, ethane, hydrogen chloride, hydrogen sulfide, ethyne]
This gas has a characteristic rotten egg smell.
Write an equation for the reaction of hydrochloric acid on copper oxide.
How will the action of dilute hydrochloric acid enable you to distinguish between sodium carbonate and sodium sulphite?
Convert two soluble metallic nitrates to insoluble metallic chlorides using dil. HCl.
State the composition of aqua regia. State which component is the oxidizing agent in aqua regia.
Name the following:
Salt obtained by heating sodium chloride with concentrated sulphuric acid below 200 degree celcius.
Fill in the blank:
On addition of silver nitrate to hydrochloric acid ___________ precipitate is formed which is soluble in ____________
Match the following:
| Column A | Column B |
| 1. A substance that turns moist starch iodide paper blue. | A. Ammonium sulphate |
| 2. A compound which release a reddish brown gas on reaction with concentrated sulphuric acid and copper turnings. | B. Lead carbonate |
| 3. A solution of this compound gives dirty green precipitate with sodium hydroxide. | C. Chlorine |
| 4. A compound which on heating with sodium hydroxide produces a gas which forms dense white fumes with hydrogen chloride. | D. Copper nitrate |
| 5. A white solid which gives a yellow residue on heating | E. Ferrous sulphate |
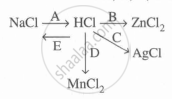
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.