Advertisements
Advertisements
प्रश्न
State the reason for the addition of caustic alkali to bauxite ore during the purification of bauxite.
Advertisements
उत्तर
Naturally, Bauxite is not soluble in normal solvents. Therefore the addition of caustic alkali to bauxite plays an important role while the extraction of aluminum. Caustic alkali dissolves bauxite forming soluble sodium meta aluminate while impurities remain insoluble and precipitate as red mud.
\[\ce{\underset{\text{(Bauxite)}}{Al2O3}.2H2O + 2NaOH -> \underset{\text{(Sodium meta aluminate)}}{2NaAlO2 + 3H2O}}\]
APPEARS IN
संबंधित प्रश्न
With a labelled diagram, explain the electro-refining of a particular metal.
`Cu (OH)2` ................................
................................
`HgS + O_2`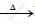 ..................
..................
Which metal is used for:
making face creams
A compound that is added to lower the fusion temperature of the electrolytic bath in the extraction of aluminium.
What do you observe when hydrogen is passed over heated copper oxide?
Write the molecular formulae of the following compound.
Copper pyrite
Write the molecular formulae of the following compound.
Ferrous tungstate
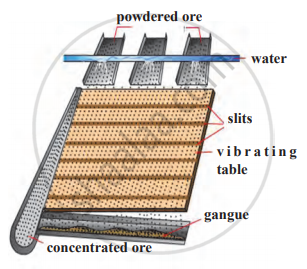
Identify the following method of concentration of ores and explain briefly.
In leaching of alumina from bauxite by Bayer's process, then ore is treated with ______.
