Advertisements
Advertisements
प्रश्न
State the following:
The group to which the element with an electronic configuration of 2, 8, 2 belongs.
Advertisements
उत्तर
Element [2, 8, 2] belongs to group 2 or [II – A]
APPEARS IN
संबंधित प्रश्न
Write the number of periods the Modern Periodic Table has. State the changes in valency and metallic character of elements as we move from left to right in a period. Also state the changes, if any, in the valency and atomic size of elements as we move down a group.
Which element has a total of two shells, with three electrons in its valence shell?
Why does the size of the atoms progressively become smaller when we move from sodium (Na) to chlorine (Cl) in the third period of the periodic table?
The elements A, B, C, D and E have atomic numbers 9, 11, 17, 12 and 13 respectively. The pair of elements which belongs to the same group of the periodic table is:
(a) A and B
(b) B and D
(c) A and C
(d) D and E
What do you understand by periodicity?
An element X belong to 4th period and 17th group, state.
name of the element.
Fill in the blank:
on moving from right to left in the second period, the number of valence electrons……….
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 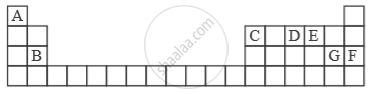
The ion of which element will migrate towards the cathode during electrolysis?
Name and state the following with reference to the elements of the first three periods of the periodic table.
Alkaline earth metal in period 3.
Name or state following with reference to the element of the first three periods of the periodic table.
A non-metal in period 2 having electronic configuration 2, 6.
